USMLE Blog
USMLE Step 1 High‐Yield Immunology Topics


If you want our team’s expert help with your USMLE prep, click here
Immunology
High Yield USMLE Step 1 Notes
Welcome to the Immunology High-Yield Notes for Step 1.
These are the most commonly tested topics on the exam—ideal for quick review in your last 2 weeks before the exam, when you’re short on time, or when you’re just starting out and want to focus on what is high yield. Created and peer-reviewed by physicians who scored 260+ on Step 1 in a Q&A style to reinforce active learning.
How to use this file
- Read the question ➔ cover the answer ➔ self-quiz before revealing.
- Review the NOTES ➔ they include high-yield details you don’t want to miss.
- Need tutoring? The Match Guy USMLE Tutoring.
- Don’t forget to watch the video lessons that go with this file. Learn more here.
Table of Contents
Spleen & Thymus
1. A histologic section of lymphoid tissue shows lymphoid follicles and surrounding periarteriolar regions. Immune cells predominate in each of these areas?
➔ B cells → Follicles (Proliferate in bacterial infections)
- Note: T cells → Periarteriolar lymphoid sheaths (PALS) (Proliferate in viral infections)
- White Pulp: B cells (Follicles), T cells (PALS).
- Marginal Zone: Macrophages, APCs.
2. A patient with a history of sickle cell disease presents with recurrent infections. What type of pathogens is he most at risk for?
➔ Encapsulated organisms
- Note: Post splenectomy/sickle cell disease (auto splenectomy) → Dec IgM → Dec C3b → Dec opsonization → Inc risk from encapsulated bacteria (SHIN) → Meningococci, H. Influenza, Pneumococci
3. A trauma patient who had a splenectomy 6 months ago. His peripheral blood smear shows small, dense, basophilic inclusions within RBCs. Likely finding?
➔ Howell-Jolly bodies
- Note: Postsplenectomy PS findings:
1. Howell-Jolly bodies (dark purple nuclear remnants)
2. Target cells (bulls-eye shaped RBCs)
4. A biopsy reveals immature lymphocytes in a mediastinal mass from a patient with fatigable weakness that improves with rest. Dx?
➔ Thymoma associated with myasthenia gravis
- Note: Myasthenia gravis is caused by anti-AChR antibodies, impairing neuromuscular transmission and causing fatigable weakness that improves with rest.
- Myasthenia Sx + mediastinal mass = Remove thymus
5. A patient with recurrent infections and a TLR4 deficiency is most likely to have infections from which type of organism?
➔ Gram-negative bacteria
- Note: TLRs recognize various PAMPs like LPS, flagellin, and nucleic acids on Gram-negative bacteria to trigger immune responses. TLR4 deficiency impairs defense against gram-negative infections.
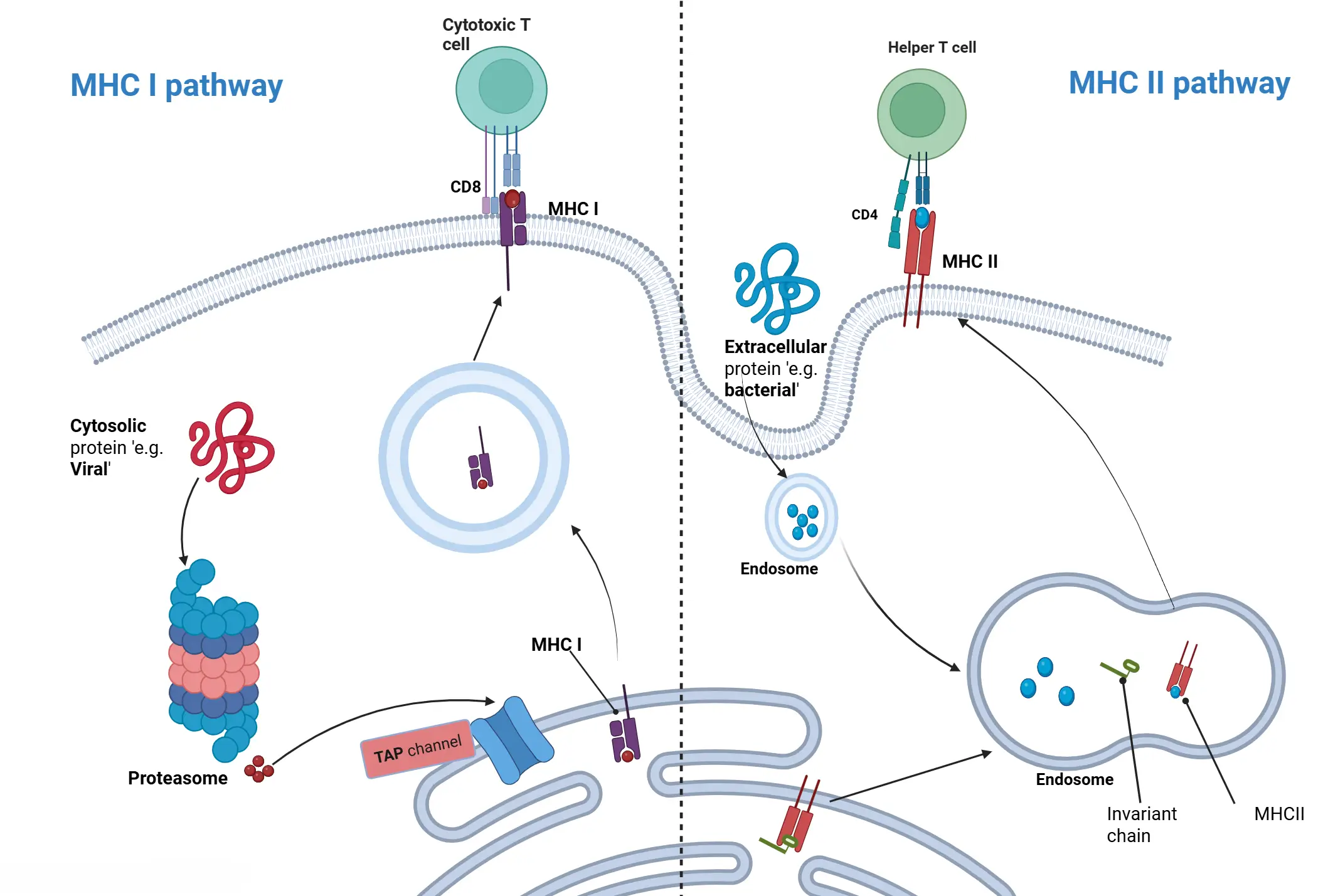
6. A 28-year-old patient with recurrent viral infection. MHC class most likely to be impaired?
➔ MHC I, presents endogenous antigens (e.g., viruses)
7. A 45M with recurrent fungal infections shows a defect in the immune response to exogenous pathogens. Which MHC class is most likely impaired?
➔ MHC II, as it presents exogenous antigens (like fungi) after processing in acidified endosomes
| Feature | MHC I | MHC II |
|---|---|---|
| Loci | HLA-A, HLA-B, HLA-C (1 letter) | HLA-DP, HLA-DQ, HLA-DR (2 letters) |
| Binds to | CD8+ T cells | CD4+ T cells |
| Antigen Type | Endogenous (viruses, tumors) – Ubiquitin-proteasome system | Exogenous (bacterial, fungi and toxins) |
| Expression | All nucleated cells – except RBCs | APCs only |
| Antigen Loading | RER via TAP | Acidified endosome |
8. A 30M presents with chronic low back pain and stiffness, particularly worse in the morning. Which HLA subtype is most likely associated with his condition?
➔ HLA-B27
- Note: HLA-B27 → Seronegative spondyloarthropathies (PAIR)
1. Psoriatic arthritis
2. Ankylosing spondylitis
3. IBD-associated arthritis
4. Reactive arthritis - B57 → Abacavir hypersensitivity
9. A patient with a CMV infection has decoy proteins that mimic MHC I on infected cells. Mechanism used to eliminate these infected cells?
➔ Antibody-dependent cell-mediated cytotoxicity (ADCC) using IgG
- Note: MHC I absent → Killing by NK cells
- MHC I present → Killing by CD 8 + T-cells
- Illusion of MHC I (decoy proteins e.g CMV) → Killing by ADCC (antibody-dependent cell-mediated cytotoxicity) using IgG
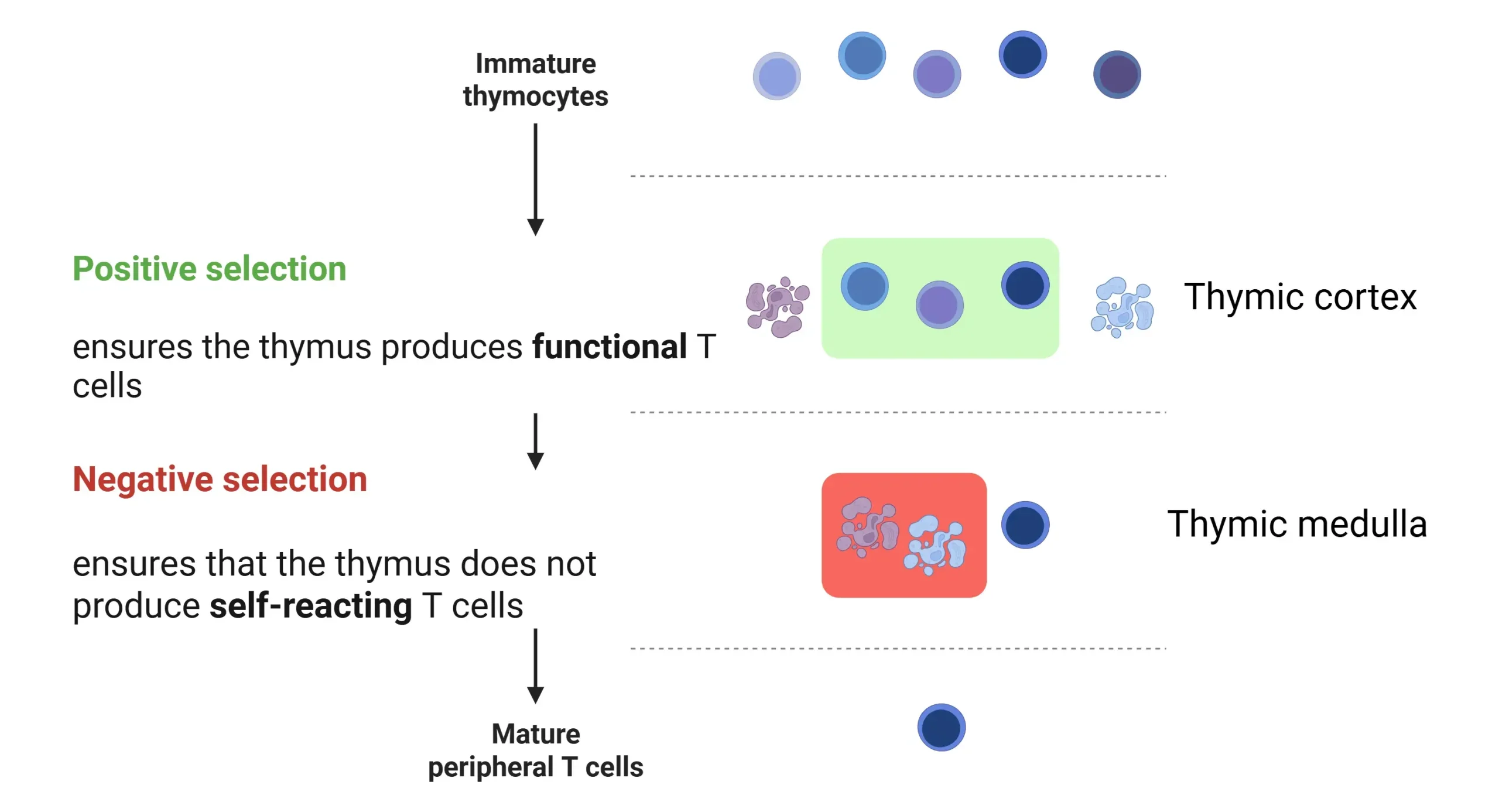
10. A 10-year-old male with DM-1,celiac disease and vitiligo has been found to have impaired T cell tolerance. Mechanism in the thymus likely disrupted?
➔ Negative selection
- Note: Impairment of negative selection (Thymic medulla) leads to improper deletion of self-reactive T cells → inc the risk of autoimmune conditions
- Positive selection occurs in the thymic cortex and ensures that T cells can recognize self-MHC molecules. T cells that cannot bind to MHC are eliminated.
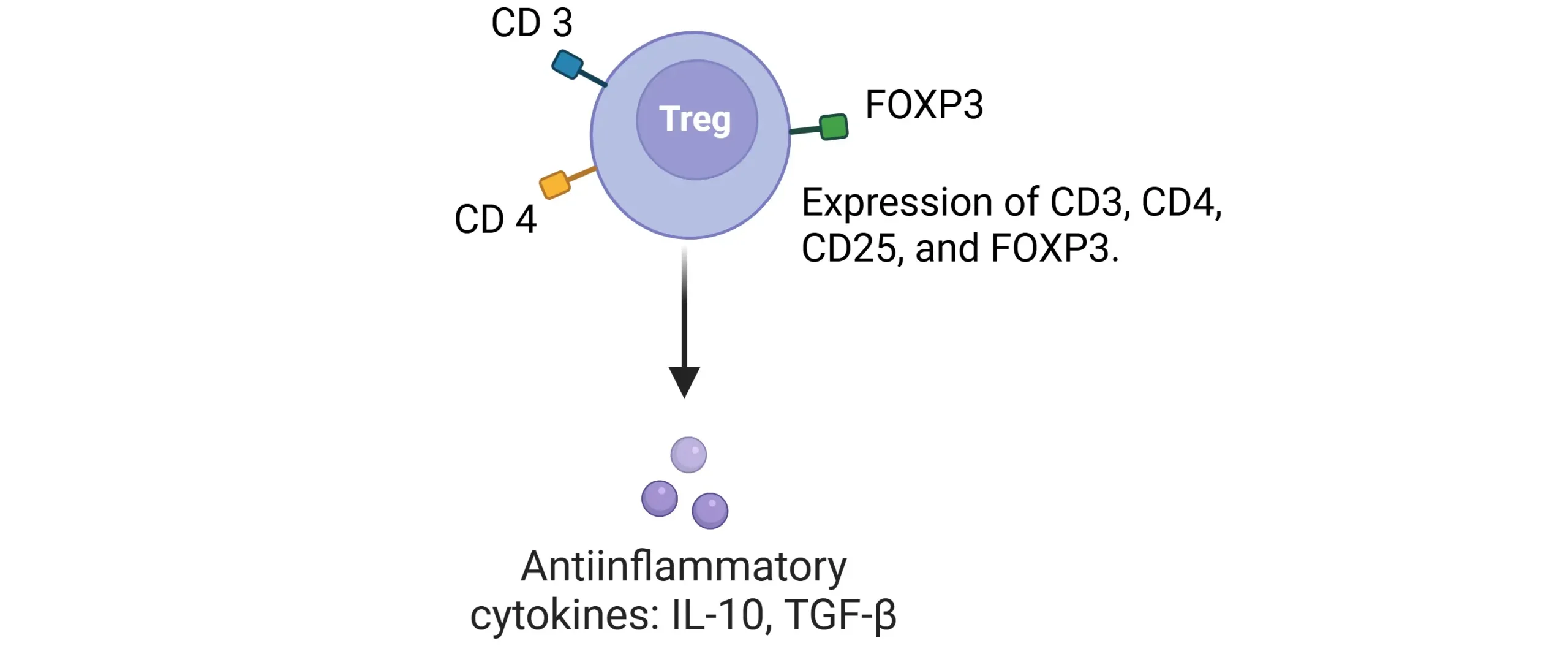
11. A 5-year-old boy presents with a history of autoimmune disease and chronic inflammation. He has been diagnosed with IPEX syndrome. Which T cell subset is most likely affected in this patient?
➔ Regulatory T cells (Tregs)
- Note: Regulatory T Cells (Tregs)
- Suppress CD4+ & CD8+ T cells to maintain immune tolerance.
- Markers: CD3, CD4, CD25, FOXP3.
- Secrete IL-10 & TGF-β (anti-inflammatory).
IPEX Syndrome (FOXP3 Deficiency) → Severe autoimmunity (X-linked).
- Immune dysregulation
- Polyendocrinopathy
- Enteropathy (severe diarrhea)
- X-linked (affects male infants, assoc. w/ diabetes).
12. A 28M with a history of recurrent viral infections which cell line is likely decreased?
➔ CD8+ T cells
- Note: CD8+ T cells are responsible for killing virus-infected and Tumor cells using Perforin, Granzyme & Fas-FasL mechanism; their deficiency impairs viral control.
Immunoglobulins
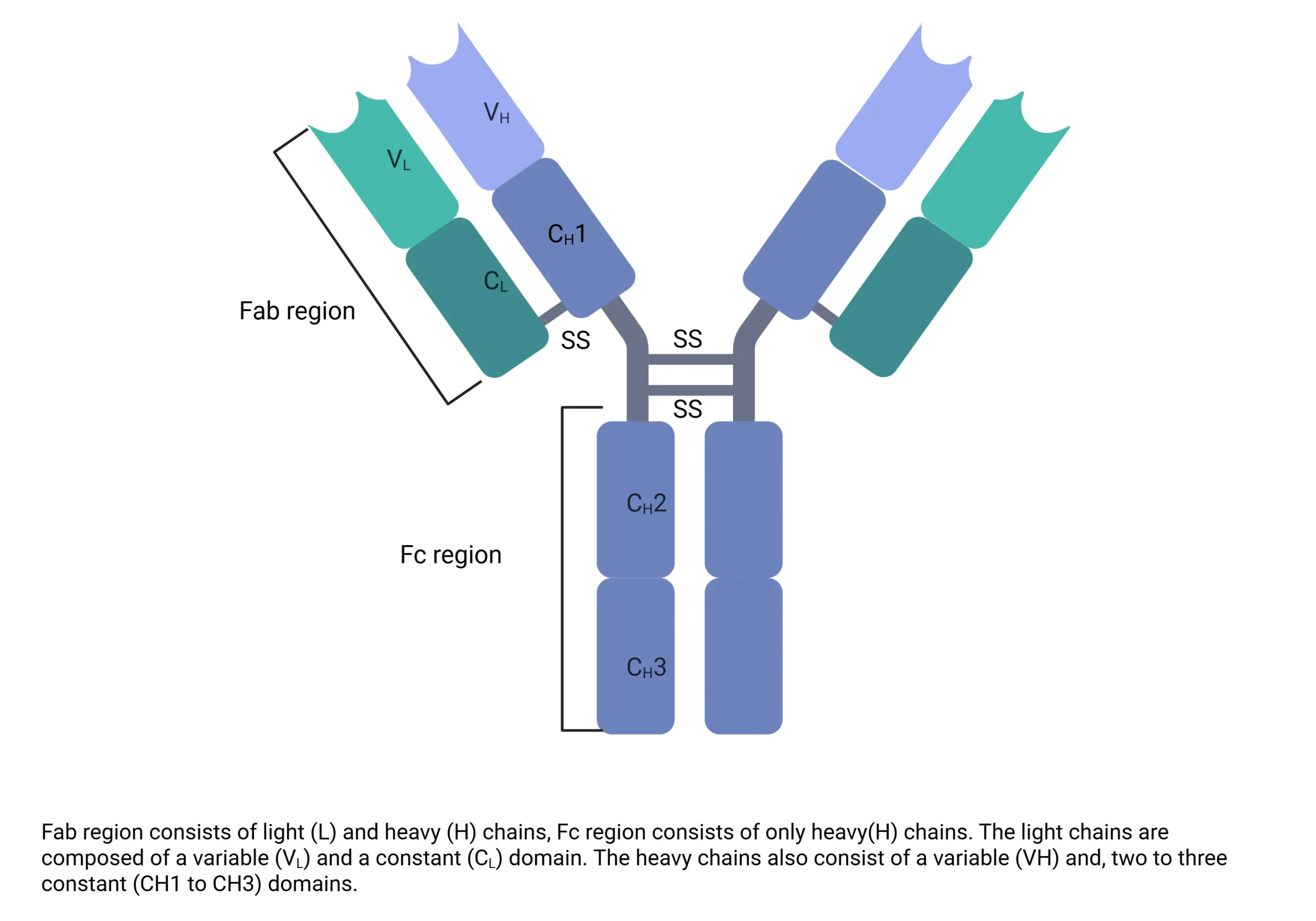
13. Patients with bacterial infection produce IgG for opsonization and complement activation. Mutation in the constant region impairs which function?
➔ Fc receptor interaction → complement fixation and opsonization
- Note: Fab (Variable Region): Binds antigen (VDJ/VJ recombination → diversity)
- Fc (Constant Region): Defines isotype (IgG, IgA, IgM), fixes complement (IgM, IgG), binds macrophages for opsonization.
14. A 6-month-old infant with recurrent infections has low serum levels of a specific immunoglobulin. Which immunoglobulin, transferred from the mother, is likely deficient?
➔ IgG
- Note: Most abundant
- Crosses placenta
- Involved in ADCC.
15. A patient with recurrent sinus infections has low levels of immunoglobulin found in mucosal secretions like tears and saliva. Which isotype is deficient?
➔ IgA
- Note: IgA provides Mucosal immunity, secreted in tears, saliva, breast milk, resistant to proteases.
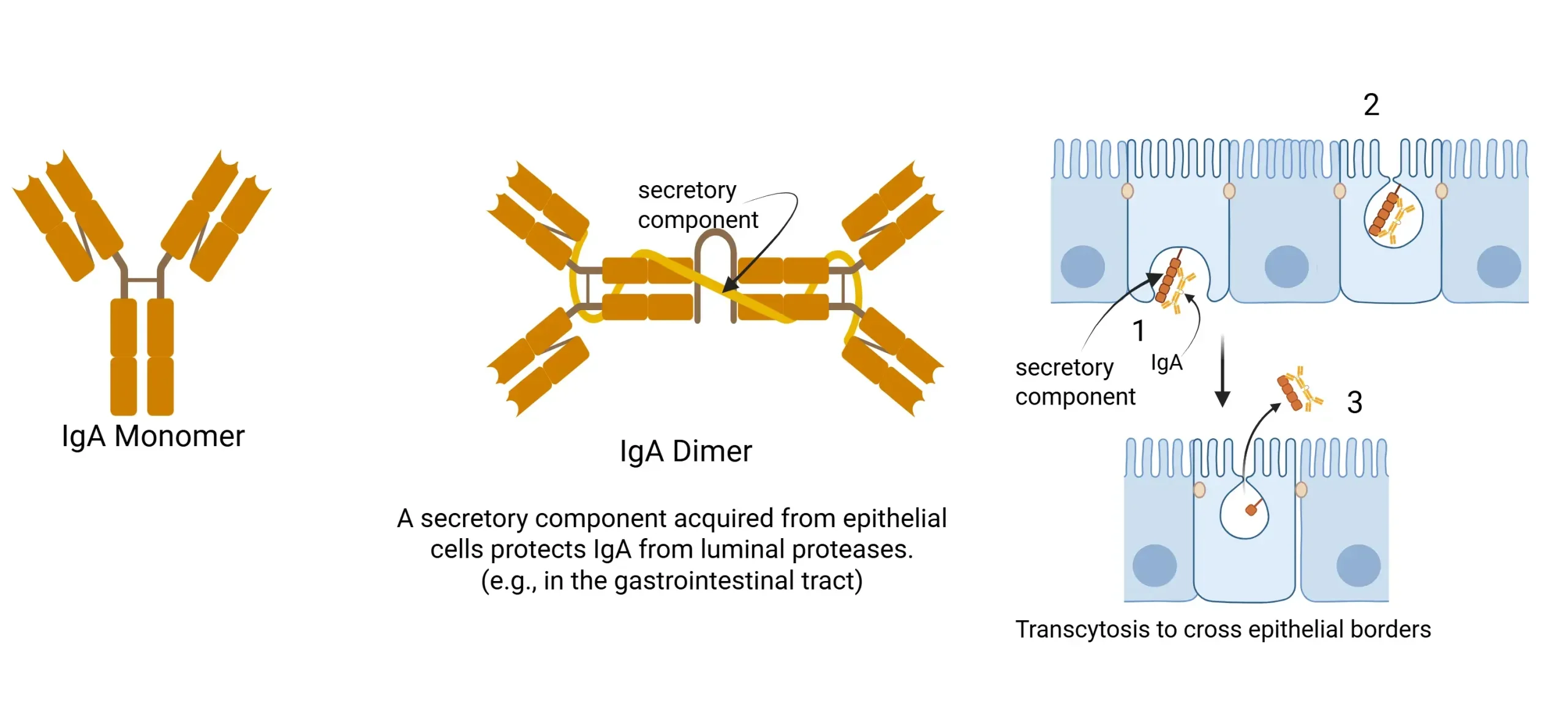
16. A patient with a recent bacterial infection has a high level of a pentameric immunoglobulin that strongly fixes complement. Which isotype is elevated?
➔ IgM
- Note: First responder in acute infections, pentameric structure, strong complement fixer.
17. A 25-year-old patient presents with a 2-week history of cough,SOB, and fever. Lab shows cold-induced hemolysis with a positive Coombs test. Immunoglobulin most likely involved?
➔ IgM – Cold autoimmune hemolytic anemia, reacts to cold temperatures
- Note: Cold-induced hemolysis is often associated with Mycoplasma pneumoniae or infectious mononucleosis (EBV).
- Warm-induced hemolytic anemia: Typically caused by IgG antibodies, commonly seen SLE or CLL.
Complement system
18. A 25M with recurrent pyogenic infections like Streptococcus and Staphylococcus presents with a family Hx of immune deficiency. Which complement component is most likely deficient?
➔ C3
- Note: C3 deficiency → Impaired opsonization → Increased susceptibility to pyogenic infections also inc risk of Immune complex diseases (SLE, glomerulonephritis).
Clinical Correlations
- C3 deficiency → Recurrent pyogenic infections (Strep, Staph), immune complex diseases (SLE, GN).
- C5-C9 deficiency → Neisseria infections (meningitis, gonorrhea).
- CD55 (DAF) deficiency → Paroxysmal Nocturnal Hemoglobinuria (PNH).
- C1 esterase inhibitor deficiency → Hereditary angioedema (bradykinin-mediated, worsened by ACE inhibitors).
19. A 40F presents with episodes of dark-colored urine in the morning, fatigue, and abdominal discomfort. Flow cytometry reveals CD55 deficiency. Dx?
➔ Paroxysmal Nocturnal Hemoglobinuria (PNH)
- Note: PNH caused by CD55 deficiency → Uncontrolled complement activation → Red blood cell lysis (especially at night).
- PNH inc the risk of thrombosis (budd-chiari syndrome)
20. A 55F with a history of recurrent episodes of swelling in the lips and tongue Sx worsened after taking antihypertensive. Drug likely taken?
➔ ACE inhibitors (Inc bradykinin levels)
- Note: C1 esterase inhibitor deficiency → Increased bradykinin levels → recurrent episodes of angioedema worsened by ACE inhibitors.
- ARBs are safe and don’t cause angioedema.
Cytokines function
21. A 55-year-old male with fever, hypotension, and signs of septic shock is found to have elevated ESR and CRP. Cytokine primarily responsible?
➔ TNF-α
- Note: First and most potent and produced by Macrophages
- WBC recruitment, vascular leak, cachexia
- Involved in Septic shock, cancer cachexia
- Other cytokines involved in acute inflammation are IL-1 & IL-6
22. A 45-year-old woman with Crohn’s disease is set to begin infliximab therapy. IGRA is positive, but chest X-ray shows no active disease. Best next step before starting treatment?
➔ Latent TB treatment before initiating infliximab
- Note: TNF-α maintains granuloma; anti-TNF (e.g., infliximab) disrupts it → TB reactivation risk.
- Latent TB Testing: IGRA (preferred in BCG-vaccinated) or TST.
- Treat first with either Isoniazid (9 months) or Rifampin (4 months).
23. A 34M with recurrent viral infections has elevated levels of a cytokine that suppresses Th1 responses. Which cytokine is responsible?
➔ IL-10
- Note: Produced by Th2 cells & Tregs
- Anti-inflammatory, suppresses Th1 response
- Overexpression of IL-10 → Susceptibility to chronic viral infections
| Cytokine | Produced by | Function | Clinical Relevance |
|---|---|---|---|
| IL-1 & IL-6 | Macrophages | Fever, acute inflammation, acute-phase protein production | Sepsis (fever, shock), inflammatory diseases (RA, COVID-19 cytokine storm) |
| TNF-α | Macrophages | WBC recruitment, vascular leak, cachexia | Septic shock, cachexia in malignancy, granuloma maintenance in TB |
| IL-8 | Macrophages | Neutrophil chemotaxis | “Clean up on IL-8” → Pus formation, infections |
| IL-12 & IFN-γ | Macrophages, Th1, NK cells | Macrophage activation, granuloma formation | Deficiency → Susceptibility to TB & mycobacterial infections |
| IL-2 | T cells | Stimulates T & NK cell growth | Used in immunotherapy (melanoma, RCC) |
| IL-4 & IL-5 | Th2 cells | IgE & IgG class switching (IL-4), IgA & eosinophil activation (IL-5) | Asthma, allergies, eosinophilia in parasitic infections |
| IL-10 | Th2 & Tregs | Anti-inflammatory, suppresses Th1 response | Overexpression → Increased susceptibility to chronic infections |
- IL-10, IL- 4, TGF-β → Anti-inflammatory → Attenuates immune response
- IL-1, IL-6, TNF-alpha → Acute inflammation
24. A 22-year-old male with persistent EBV infection and lymphadenopathy shows abnormal B-cell proliferation. Which B-cell marker is targeted by EBV?
➔ CD21
- Note: CD21 is the receptor for EBV on B cells.
- Essential for EBV entry into B cells, leading to Infectious Mononucleosis
25. A patient with non-Hodgkin lymphoma undergoes flow cytometry, revealing clonal expansion of B cells. Which markers would be most prominently expressed?
➔ CD19, CD20
- Note: CD19 and CD20 are key B-cell markers involved in B-cell activation and proliferation.
- CD20 is a therapeutic target for rituximab, commonly used in B-cell lymphomas.
Vaccines
26. A 4-year-old child with bruton agammaglobulinemia is scheduled for routine vaccinations. Which polio vaccine should be avoided?
➔ Oral Polio Vaccine (Sabin)
- Note: Live Attenuated Vaccines: MMR, Varicella, Sabin, Yellow fever → Risk of reversion → Avoid in pregnancy, immunocompromised (HIV CD4 < 200, SCID, agammaglobulinemia)
- Salk Polio (Inj) is Killed and safe for immunocompromised patients
27. A 40 M recovering from a puncture wound presents to the clinic 4 days later with muscle stiffness and spasms. He recalls not getting a booster shot in over 10 years. What is the immediate intervention?
➔ Tetanus toxoid vaccine + Tetanus Immune Globulin (TIG)
- Note: Toxoid → Long-term protection
- TIG → Immediate passive immunity
- Clean wound → Toxoid if >10 years
- Dirty wound → Toxoid + TIG if >5 years or unknown status
| Live Attenuated | Strong cellular & humoral response. Long-lasting, but avoid in pregnancy & immunodeficiency due to reversion risk. | MMR, Varicella, Oral polio, Yellow fever “Live! MVSY” (MMR, Varicella, Sabin, Yellow fever) |
| Killed/Inactivated | Only humoral response needs boosters, safer than live. | Rabies, Hep A, Influenza (injected), Salk polio (Inj) “RIP Always” (Rabies, Influenza, Polio [Salk], Hep A) |
| Subunit | Only antigenic parts are used. Weaker response. | HBV (HBsAg), HPV, Meningococcus, Pneumococcus, Pertussis “HP-MPP” → Help Protect Many People” (HPV, Pneumococcus, Meningococcus, Pertussis) |
| Toxoid | Inactivates bacterial toxins, provides strong antibody-mediated immunity, but requires boosters. | “Toxic T&D” Tetanus, Diphtheria |
Hypersensitivity Reactions
28. A patient experiences sudden itching, hives, and difficulty breathing minutes after a bee sting. Which mediator is primarily responsible for these immediate symptoms?
➔ Histamine
- Note: Histamine is the first mediator released from mast cells during an allergic/anaphylactic reaction.
- Triggers vasodilation, bronchoconstriction, and increased capillary permeability → Classic signs of anaphylaxis.
- Responsible for rapid-onset symptoms like flushing, itching, and hypotension.
Type II vs Type III hypersensitivity
- Type II: Localized damage (1–2 organs) → AIHA, MG
- Type III: Systemic involvement (≥3 organs) → SLE, serum sickness
29. A patient receives a PPD skin test for tuberculosis screening. After 48 hours, a firm, raised induration is observed at the injection site. Which immune cells are driving this response?
➔ CD4+ Th1 cells
- Note: Type IV hypersensitivity
- Th1 cells release IFN-γ, activating macrophages.
- IL-12 from macrophages amplifies the Th1 response.
- This mechanism underlies PPD tests, contact dermatitis, and granuloma formation.
PPD Test & Granuloma Formation:
- Mycobacterial antigen → activates Th1 cells → release IFN-γ → macrophage activation → formation of epithelioid cells and giant cells → granuloma develops → maintained by TNF-α and CXCL chemokines.
| Type | Mechanism | Key Mediators | Examples |
|---|---|---|---|
| Type I (Immediate, IgE) | IgE → Mast cell degranulation → Histamine, Leukotrienes | Histamine, Tryptase (marker), LTC₄, LTD₄, LTE₄ | Anaphylaxis, Allergic asthma |
| Type II (Antibody-mediated, IgG/IgM) | Antibodies bind cell antigens → Destruction, Inflammation, Dysfunction | IgG, IgM, C3b (opsonization), C5a (inflammation) | Destruction: AIHA, ITP, Transfusion reaction Inflammation: Goodpasture, Rheumatic fever Dysfunction: Myasthenia gravis, Graves |
| Type III (Immune complex, IgG) | Immune complexes deposit → Complement activation → Neutrophil damage | Immune complexes, ↓ C3, ↓ C4 | SLE, RA, PSGN, Serum sickness-like reactions from some drugs such as penicillins and monoclonal antibodies |
| Type IV (Delayed, T-cell mediated) | CD8+ T cells: Direct killing CD4+ T cells: IFN-γ → Macrophage activation → TNF-α | T cells, IFN-γ, TNF-α | Contact dermatitis, TB test (PPD), GVHD, Multiple Sclerosis |
Transfusion Reactions
30. A patient undergoing a blood transfusion develops fever, flank pain, hemoglobinuria, and hypotension within the first hour. What type of hypersensitivity is involved?
➔ Type II hypersensitivity (Acute Hemolytic Transfusion Reaction)
- Note: During or within 24 hours of transfusion
- Caused by ABO incompatibility → Complement-mediated lysis
- Presents with fever, hemoglobinuria, jaundice, hypotension & flank pain
| Reaction Type | Cause | Key Clinical Feature |
|---|---|---|
| Allergic/Anaphylactic Reaction → Minutes to 2-3 hrs | Type I hypersensitivity to plasma proteins (IgA in IgA-deficient) | Urticaria, pruritus, wheezing, hypotension, shock |
| Acute Hemolytic Transfusion Reaction → During transfusion or within 24 hrs | Type II hypersensitivity (ABO incompatibility), Mainly by IgM | Fever, hypotension, hemoglobinuria, jaundice, flank pain. |
| Febrile Non-Hemolytic Reaction → 1-6 hrs | Cytokines from donor WBCs | Fever, chills, flushing, headache |
| Transfusion-Related Acute Lung Injury (TRALI) → Minutes to 6 hrs | Recipient Neutrophil against donor antibodies | Respiratory distress, non-cardiogenic pulmonary edema |
| Delayed Hemolytic Reaction → Onset over 24 hrs, usually 1-2 weeks | Anamnestic response to minor RBC antigens | Mild fever, hyperbilirubinemia, clinically silent |
Immunosuppressants
31. A 45-year-old male undergoes a kidney transplant and is started on an immunosuppressant. A few months later, he developed nephrotoxicity and gingival hyperplasia. Drug responsible?
➔ Cyclosporine
- Note: Cyclosporine is a calcineurin inhibitor → ↓ IL-2 transcription → ↓ T-cell activation.
- Nephrotoxicity + gingival hyperplasia = Cyclosporine toxicity.
- Nephrotoxicity + diabetes = Tacrolimus toxicity.
- Tacrolimus is a calcineurin inhibitor, binds FKBP → ↓ IL-2 transcription.
- Causes nephrotoxicity & ↑ risk of diabetes.
32. A 58-year-old woman with a history of kidney transplantation is on Sirolimus (Rapamycin) as part of her immunosuppressive regimen. What is the MOA of this drug?
➔ mTOR inhibitor → Blocks T & B cell activation by reducing their response to IL-2
- Note: Sirolimus is unique for being non-nephrotoxic.
- Primarily used for kidney transplant prophylaxis.
33. A patient receiving immunosuppressive therapy after an organ transplant develops tremors. His medication is a monoclonal antibody that blocks IL-2 receptors.Drug responsible?
➔ Basiliximab
- Note: Basiliximab is a Monoclonal antibody → Blocks IL-2R
- Side Effect: Tremor
34. Which drug is a monoclonal antibody against complement protein C5 and is used to treat paroxysmal nocturnal hemoglobinuria (PNH)?
➔ Eculizumab
| Drug | Target | Disease Treated |
|---|---|---|
| Adalimumab | TNF-α | Rheumatoid arthritis, Crohn’s disease |
| Infliximab | TNF-α | Crohn’s disease, Ulcerative colitis |
| Eculizumab | Complement C5 | Paroxysmal nocturnal hemoglobinuria (PNH) |
| Denosumab | RANKL | Osteoporosis |
| Omalizumab | IgE | Severe asthma |
| Rituximab | CD20 (B cells) | Non-Hodgkin lymphoma, RA, CLL |
| Trastuzumab | HER2/neu | HER2-positive breast cancer |
| Bevacizumab | VEGF | Colorectal cancer, lung cancer |
35. A patient with relapsing-remitting multiple sclerosis is being treated with interferon beta. What is the primary immune target of this drug?
➔ T cells (specifically reduces Th1 proinflammatory activity)
- Note: IFN-β reduces Th1 T-cell activity and inflammation.
- Used to treat relapsing-remitting MS.
- Has antiviral and immunomodulatory effects.
36. A leukemia patient on azathioprine develops severe pancytopenia after starting a newer drug for gout. Drug likely responsible?
➔ Allopurinol
- Note: Azathioprine is a 6-MP prodrug → Inhibits nucleotide synthesis → lymphocyte proliferation.
- Allopurinol inhibits xanthine oxidase (XO), increasing azathioprine levels (also metabolized by XO) and increasing the risk of pancytopenia.
Immunological Disorders
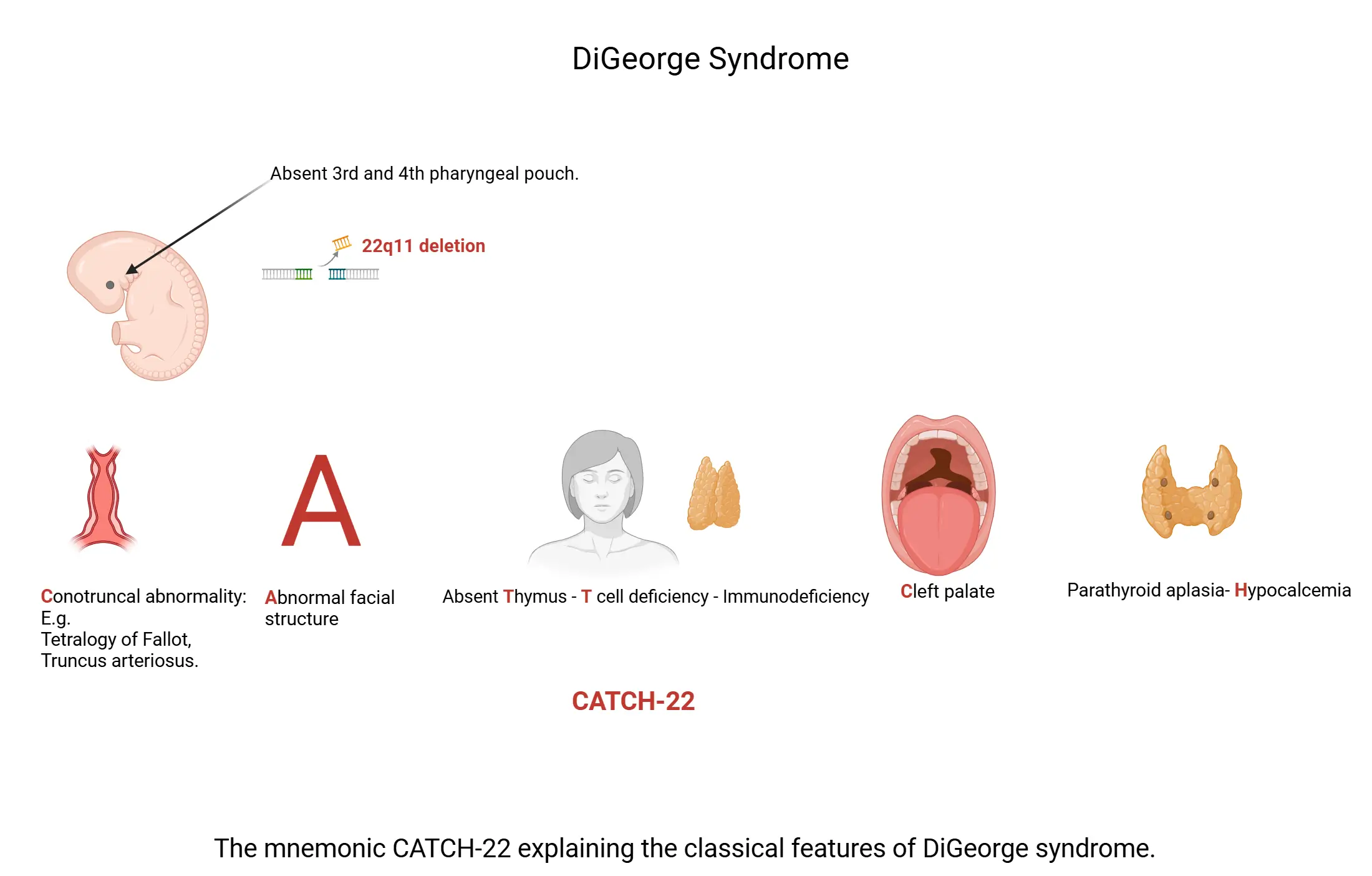
37. A newborn with facial dysmorphia, hypocalcemia, and a harsh systolic murmur undergoes a chest X-ray revealing an absent thymic shadow. What laboratory findings would you expect?
➔ ↓ T cells, ↓ PTH, ↓ Calcium
- Note: DiGeorge Syndrome (22q11 deletion) → Defective 3rd pharyngeal pouch
- Absent thymus & parathyroids → Impaired T-cell production & hypocalcemia
- Presents with congenital heart defects (TOF, TGA) recurrent viral/fungal infections and hypocalcemia
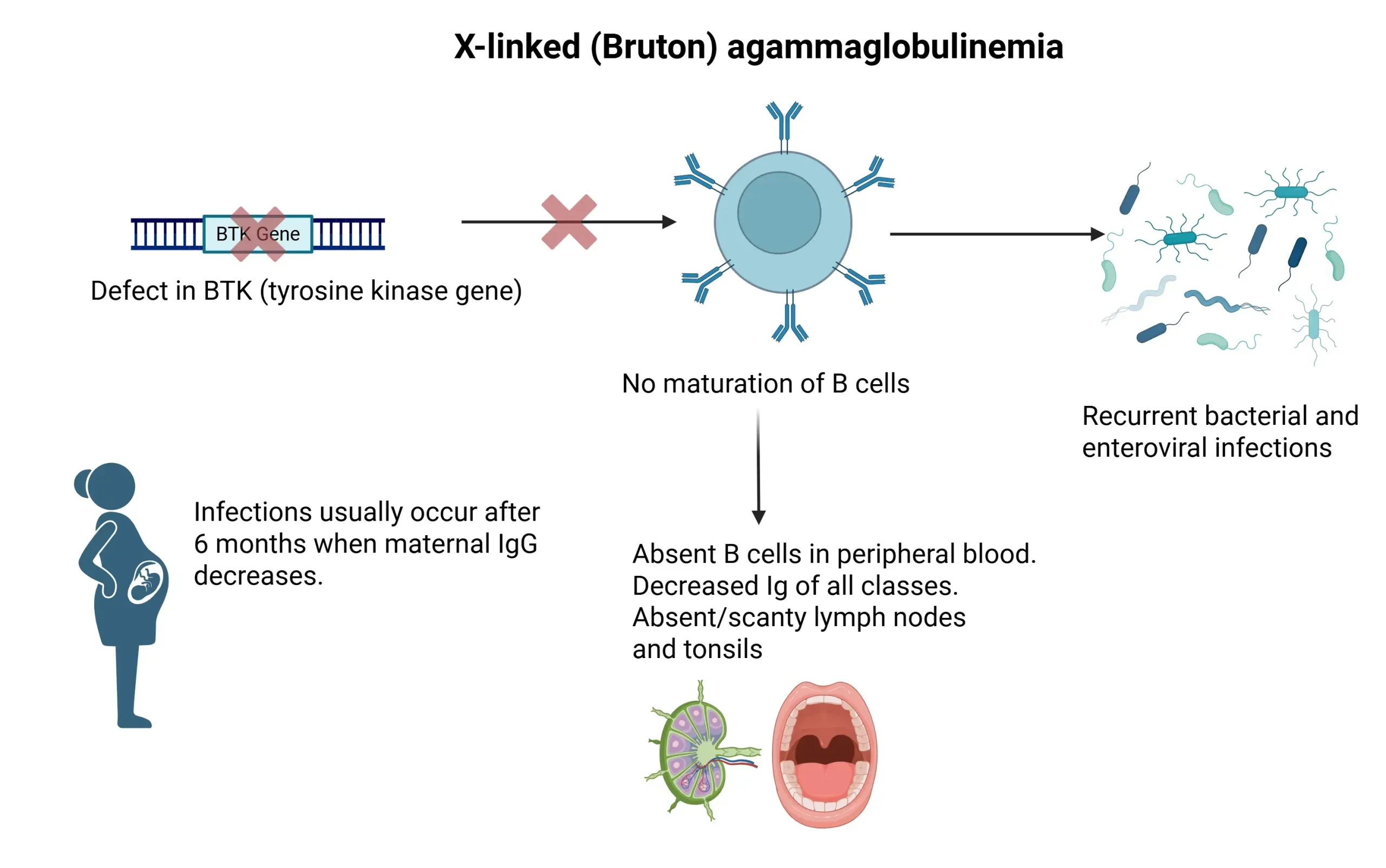
38. An infant is diagnosed with Bruton Agammaglobulinemia. What would you expect in a lymph node biopsy?
➔ Absence of germinal centers and primary follicles
- Note: X-linked (Bruton) Agammaglobulinemia → No mature B cells → No germinal center formation.
- High risk of encapsulated bacterial infections (SHiN organisms)
39. A 3-month-old boy presents with recurrent viral, bacterial, and fungal infections along with chronic diarrhea and failure to thrive. Labs reveal absent T & B cells. Dx?
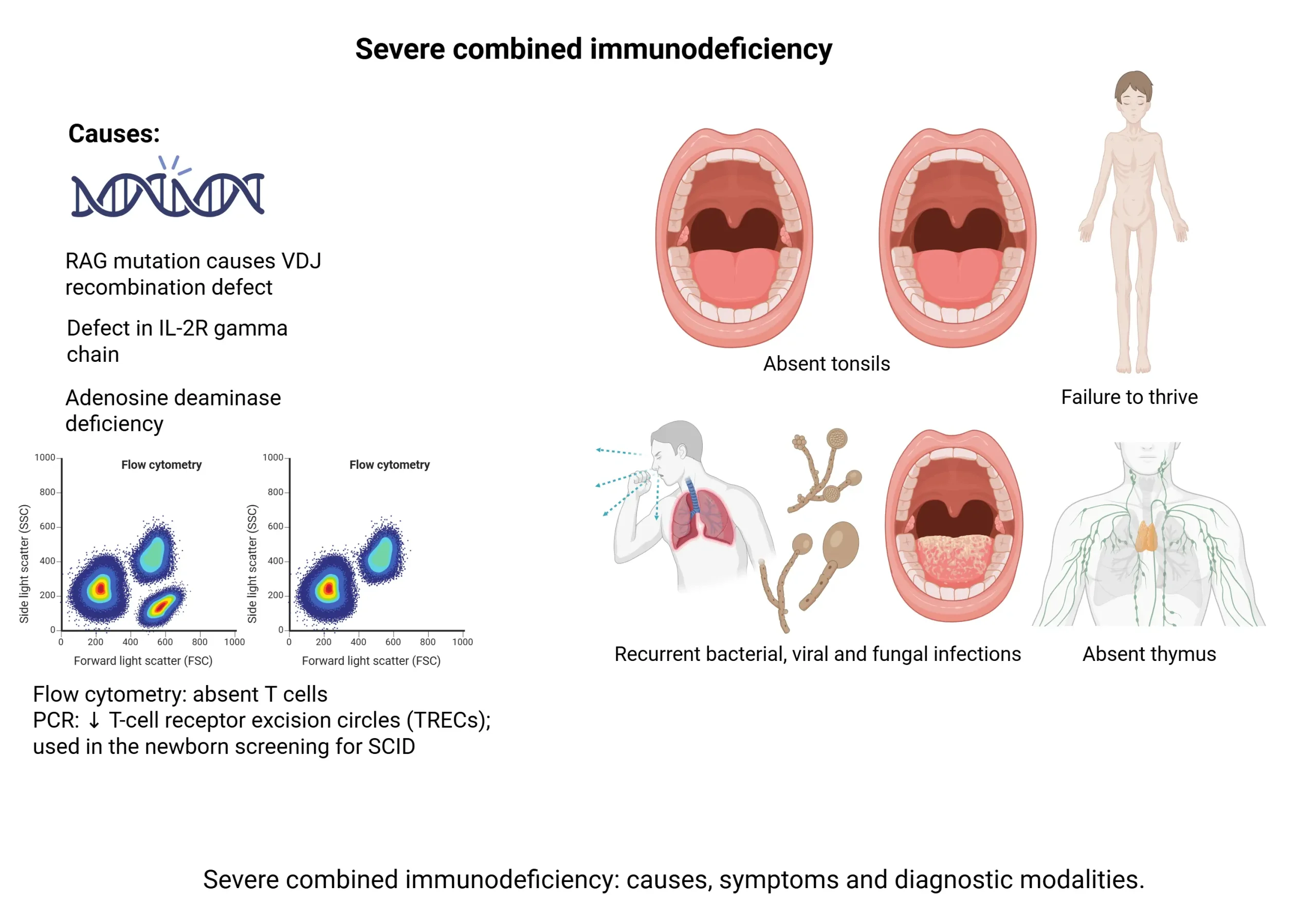
➔ Severe Combined Immunodeficiency (SCID)
- Note: SCID → IL-2Rγ mutation (X-linked) or ADA deficiency (Autosomal Recessive) → No T Cells (absent T-cell surface protein- CD-3,CD-28) as a result no B-cells too.
- Labs: ↓ TRECs (T-cell receptor excision circles), absent thymic shadow, no germinal centers
40. A 7M with recurrent skin and soft tissue abscesses from S. aureus is diagnosed with a condition where his phagocytes fail to generate reactive oxygen species. Diagnostic test to confirm?
➔ DHR (Dihydrorhodamine) flow cytometry test → ↓ green fluorescence
- Note: Chronic Granulomatous Disease (CGD) caused by NADPH oxidase deficiency → Impaired respiratory burst.
| Disorder | Pathogenesis | Clinical Features | Key Findings |
|---|---|---|---|
| B-Cell Disorders | |||
| X-linked (Bruton) Agammaglobulinemia (XR) | BTK mutation → No B-cell maturation | Recurrent bacterial, Giardia & enteroviral infections after 6 months | ↓ B cells, ↓ IgG, IgA, IgM, Absent lymph nodes/tonsils |
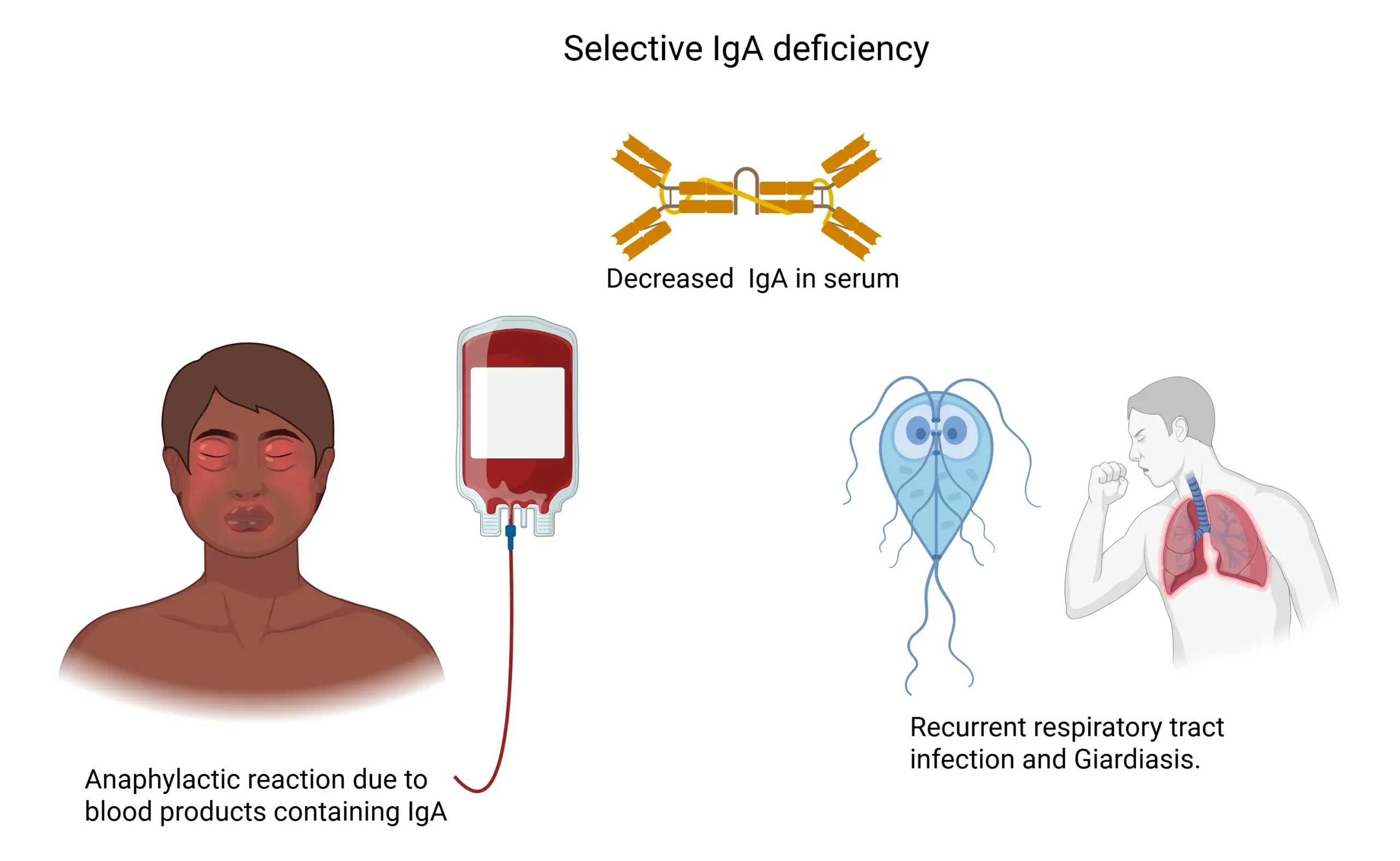
| Selective IgA Deficiency | Defective IgA production | Mostly asymptomatic; ↑ risk of GI, airway infections, anaphylaxis to blood products | ↓ IgA, Normal IgG/IgM, False-negative celiac test |
| T-Cell Disorders | |||
| DiGeorge Syndrome (22q11) | 3rd & 4th pharyngeal pouch defect → Absent thymus & parathyroids | Viral/fungal infections, tetany (hypocalcemia), TOF, TGA | ↓ T cells, ↓ PTH, ↓ Ca²⁺, Absent thymic shadow |
| Hyper-IgE Syndrome (Job Syndrome, AD) | STAT3 mutation → Impaired Th17 function → ↓ Neutrophil recruitment | Cold staph abscesses, coarse facies, retained primary teeth, fractures, eczema | ↑ IgE, ↑ eosinophils |
| Combined B- and T-Cell Disorders | |||
| Severe Combined Immunodeficiency (SCID) (XR/AR) | IL-2Rγ mutation (XR) or ADA deficiency (AR) → No T/B cells | Failure to thrive, chronic diarrhea, recurrent infections (all types) | ↓ TRECs, Absent thymic shadow, germinal centers, T-cells |
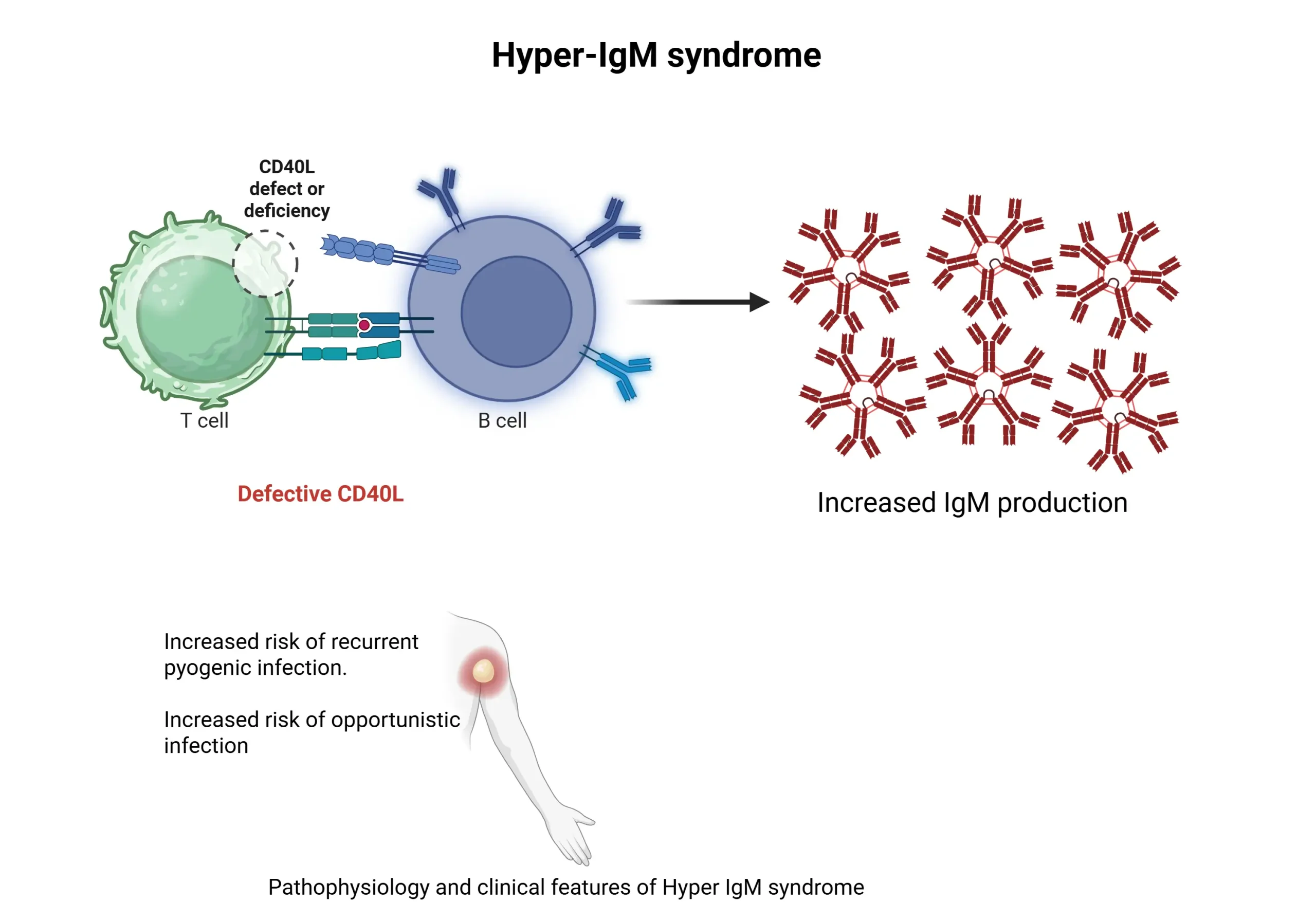
| Hyper-IgM Syndrome (XR) | CD40L defect → No class switching | Severe pyogenic infections, PCP, CMV, Cryptosporidium | ↓ IgG, IgA, IgE, Normal IgM |
| Phagocyte Disorders | |||
| Leukocyte Adhesion Deficiency (LAD-1) (AR) | CD18 defect (LFA-1 integrin) → Impaired neutrophil migration | Delayed umbilical cord separation, recurrent non-purulent infections | Marked neutrophilia |
| Chronic Granulomatous Disease (CGD) (XR) | NADPH oxidase deficiency → ↓ ROS & respiratory burst | Recurrent skin abscesses, osteomyelitis, granulomas | Catalase+ organisms (S. aureus, Serratia, Nocardia, Aspergillus), DHR test: ↓ green fluorescence |
- Recurrent infections from Catalase-positive (S. aureus, Serratia, Burkholderia, Nocardia, Aspergillus)
Types of graft rejection
41. A kidney transplant recipient shows a sudden rise in creatinine and biopsy reveals lymphocytic infiltration and C4d deposition. What is the underlying immune mechanism?
➔ Acute Rejection — Mediated by Cellular (CD8+ & CD4+ T cells) and Humoral (antibodies, C4d deposition)
- Note: Findings: Vasculitis with lymphocytic infiltration
- Reversible with immunosuppression
42. A patient undergoes an allogeneic bone marrow transplant and develops a maculopapular rash, jaundice, and diarrhea 3 weeks later. Dx?
➔ Graft-versus-Host Disease (GVHD)
- Note: Caused by Donor T cells attack host tissues; common after allogeneic bone marrow & liver transplant.
- Presents with maculopapular rash, jaundice & watery diarrhea.
| Type | Cause | Immune Mechanism | Clinical Features |
|---|---|---|---|
| Hyperacute Minutes – during surgery | Pre-existing antibodies (Type II HSR) due to anti ABO or anti HLA antibodies | Antibody-mediated, complement activation | Widespread thrombosis, ischemia, necrosis; AKA ‘white graft rejection’ Graft removal required |
| Acute Weeks–Months | Donor MHC activates T & B cells | Cellular: CD8+ & CD4+ T cells (Type IV HSR) Humoral: Antibodies (C4d deposition) | Vasculitis with lymphocytic infiltrate; reversible with immunosuppression |
| Chronic Months–Years | CD4+ T cells respond to donor peptides Vascular wall thickening and luminal narrowing | Cellular & Humoral (Type II & IV HSR) | Arteriosclerosis, fibrosis, parenchymal atrophy, organ-specific failure (e.g., chronic allograft nephropathy, bronchiolitis obliterans)—Refractory to immunosuppressants |
🏁 Final Thoughts
You made it to the end—well done! These high-yield notes are designed to save you time and keep you focused on what actually matters for Step 1. Review these vignettes regularly—steady practice beats cramming.
Need one-on-one tutoring? Visit The Match Guy USMLE Tutoring — we’ve helped hundreds of students reach their score goals.
Don’t forget to watch the video lessons that go with this file. They are part of our 7-day high-yield review of Step 1.
Join this RISK FREE live bootcamp 👉 Sign up here
Good luck and we’re rooting for you all the way to Match Day! 🎯
Disclaimer
Version 1.0 May 2025. No part of this document may be reproduced or distributed without written permission. For personal study only. Medical information herein is for educational purposes and does not constitute clinical advice. Always confirm with current guidelines and your institution’s policies.
Prefer a narrative review instead of Q&A style? Scroll down to the section below for high-yield concepts in paragraph format (they cover the same concepts).
Narrative Format
High-Yield Immunology Topics
High-yield Immunology review for USMLE: Spleen, thymus, innate/adaptive immunity, MHC I/II, T/B cell activation, antibodies, complement, hypersensitivity, immunodeficiency. Ace your exam!
Table of Contents
- Spleen Structure
- Thymus
- Major histocompatibility complex I and II
- Role of Natural Killer Cells
- Differentiation of T cells
- T & B Cell Activation (High-Yield Cytokine Interactions)
- Antibody Structure
- Immunoglobulin Isotypes
- Complement
- Clinically Relevant Cytokines
- Cell surface proteins
- High-Yield Vaccines
- Hypersensitivity types
- Transfusion Reactions
- Immunosuppressants
- Immunodeficiency Disorders
- Transplant Rejection
Spleen Structure
- White Pulp: B cells (Follicles), T cells (PALS).
- Marginal Zone: Macrophages, APCs.
- Red Pulp: RBCs, removes old/damaged RBCs.
➢ Post splenectomy/sickle cell disease (auto splenectomy) → Dec IgM → Dec C3b → Dec opsonization → Inc risk from encapsulated bacteria (SHIN) → Streptococci, H. Influenza, Pneumococci
Postsplenectomy PS findings:
- Howell-Jolly bodies (dark purple nuclear remnants)
- Target cells (bullseye-shaped RBCs).
How it is tested:
A 45-year-old man had post-emergency surgery for blunt trauma six months ago. Peripheral smear shows RBCs with small, round, dark inclusions.
Answer: Howell-Jolly bodies
Thymus
- Derived from 3rd pharyngeal pouch → contain T-cells
- Absent in DiGeorge syndrome and SCID
- Neoplasmof thymus → myasthenia gravis

How it is tested:
A 28-year-old woman presents with intermittent diplopia, ptosis, and muscle weakness that worsens with use and improves with rest. On examination, her reflexes are normal, and an ice pack test improves her ptosis. What is the most likely cause of her illness?
Answer: Antibodies to acetylcholine receptors (AChR)
| Feature | Innate Immunity | Adaptive Immunity |
|---|---|---|
| Response | Rapid, nonspecific, no memory | Slow, highly specific, memory response present |
| Secreted Proteins | Complement, cytokines, CRP, defensins | Immunoglobulins |
| Pathogen Recognition | TLRs recognize PAMPs (LPS, flagellin, nucleic acids) | Via Memory cells |
Major histocompatibility complex I and II
| Feature | MHC I | MHC II |
|---|---|---|
| Loci | HLA-A, HLA-B, HLA-C (1 letter) | HLA-DP, HLA-DQ, HLA-DR (2 letters) |
| Binds to | CD8+ T cells | CD4+ T cells |
| Antigen Type | Endogenous (viruses, tumors) – Ubiquitin-proteasome system | Exogenous (bacteria, fungi, toxins) |
| Expression | All nucleated cells – except RBCs | APCs only |
| Antigen Loading | RER via TAP | Acidified endosome |
How it is tested:
A 5-year-old boy presents with recurrent skin and lung infections. His past medical history includes recurrent pneumonia and abscess formation. Laboratory tests reveal a negative nitro blue tetrazolium (NBT) test. Immune processes defective in this patient?
Answer: Chronic granulomatous disease (CGD) → Impaired acidification of lysosomes → MHC II involvement
➢ HLA-B27 → Seronegative spondyloarthropathies (PAIR)
- Psoriatic arthritis
- Ankylosing spondylitis
- IBD-associated arthritis
- Reactive arthritis
➢ B57 → Abacavir hypersensitivity
Role of Natural Killer Cells
➢ Absence of MHC I on target cell is an activation signal for NK cells
➢ NK doesn’t need thymus to function, so they can work in athymic patient
- MHC I absent → Killing by NK cells
- MHC I present → Killing by CD 8 + T-cells
- Illusion of MHC I (decoy proteins e.g CMV)→ Killing by ADCC (antibody-dependent cell-mediated cytotoxicity) using IgG
Differentiation of T cells
➢ Positive Selection (Thymic Cortex) → T cells survive if they bind self-MHC on cortical epithelial cells.
➢ Negative Selection (Thymic Medulla) → T cells die if they bind self-antigens too strongly OR Become Tregs (regulatory T cells).
○ Tolerance due to -ve selection
○ AIRE Protein (Autoimmune Regulator) → Deficiency → Autoimmune Polyendocrine Syndrome-1 (APS-1)
Regulatory T Cells (Tregs)
- Suppress CD4+ & CD8+ T cells to maintain immune tolerance.
- Markers: CD3, CD4, CD25,FOXP3.
- Secrete IL-10 & TGF-β (anti-inflammatory).
IPEX Syndrome (FOXP3 Deficiency) → Severe autoimmunity (X-linked).
- Immune dysregulation
- Polyendocrinopathy
- Enteropathy (severe diarrhea)
- X-linked (affects male infants, assoc. w/ diabetes).
How it is tested:
A 2-month-old male presents with chronic diarrhea, eczema, and failure to thrive. The lab shows elevated blood glucose levels. Genetic testing reveals a mutation affecting a transcription factor essential for Treg development. Cytokines most likely deficient?
Answer: IPEX Syndrome → IL-10, TGF-β
✔ Th1 cells → IFN-γ → Activates macrophages (↑ killing)
✔ CD40L (lymphocytes) ↔ CD40 (macrophages) interaction boosts response.
T & B Cell Activation (High-Yield Cytokine Interactions)
| Cell Type | Signal 1 (Antigen Recognition) | Signal 2 (Co-stimulation) | Notes (Cytokines & Class Switching) |
|---|---|---|---|
| T Cells (CD4+ & CD8+) | CD4+ → APC presents MHC II CD8+ → APC presents MHC I | CD80/86 (B7) on APC binds CD28 on T cell | CD4+ Th Cytokines: IL-2 (T-cell growth), IL-4 (IgE), IL-5 (IgA), IFN-γ (IgG) CD8+ Tc Function: Kills virus-infected/tumor cells (Perforin, Granzyme, Fas-FasL) |
| B Cells | BCR binds antigen → Presents on MHC II to Th | CD40 (B cell) binds CD40L (Th cell) → Class switching | Class Switching: IL-4 → IgE, IgG; IL-5 → IgA; IFN-γ → IgG (opsonization) |
How it is tested:
A 5-year-old boy with recurrent bacterial infections has normal T-cell count but defective class switching. Genetic testing shows a mutation in a costimulatory molecule.
Which interaction is most likely defective?
Answer: CD40–CD40L (Hyper-IgM Syndrome)
Antibody Structure

➢ Fab (Fragment, antigen-binding)
- Variable region → Binds antigen (unique idiotype)
In the variable portion, VDJ recombination in heavy chains and VJ recombinations lead to antibody diversity.
➢ Fc (Constant region)
- Determines the isotype (IgG, IgA, IgM, etc.)
- Fixes complement (IgM, IgG)
- Binds macrophages for opsonization
Isotype class switching occurs by splicing out DNA of heavy chain constant region until desired type is reached. The variable region and antibody specificity stay the same.
How it is tested:
A patient with a bacterial infection produces IgG antibodies that facilitate opsonization and complement activation. A mutation affecting the antibody’s constant region would most likely impair which of the following functions?
Answer: Interaction with Fc receptors on phagocytes → complement fixation and opsonization affected
Immunoglobulin Isotypes
➢ IgG– Most abundant in serum, crosses placenta, fixes complement, only one to involve in ADCC, opsonizes bacteria, secondary response provides long-term immunity (“IgG Greets the Growing fetus”)
➢ IgA– Mucosal immunity, most produced overall, found in secretions (tears, saliva, breast milk), dimer with J chain, resistant to proteases. (Secretory IgA protects surfaces)
➢ IgM– First antibody in an acute infection, pentamer in circulation, strong complement fixer, antigen receptor on naïve B cells. (“IMmediate response”)
➢ IgE:Type I hypersensitivity, binds mast cells & basophils, releases histamine, activates eosinophils (parasites), involved in ABPA (Allergic Bronchopulmonary Aspergillosis).
How it is tested:
A patient presents with recurrent sinus and respiratory infections. Laboratory analysis reveals a deficiency in an immunoglobulin isotype that forms dimers, is resistant to proteases, and is found in mucosal secretions. Which isotype is most likely deficient in this patient?
Answer: IgA
➢ Antigens without a peptide component cannot be presented by MHC to T cells, are weakly immunogenic, and often require adjuvants or booster doses in vaccines (e.g., pneumococcal polysaccharide vaccine).
Complement
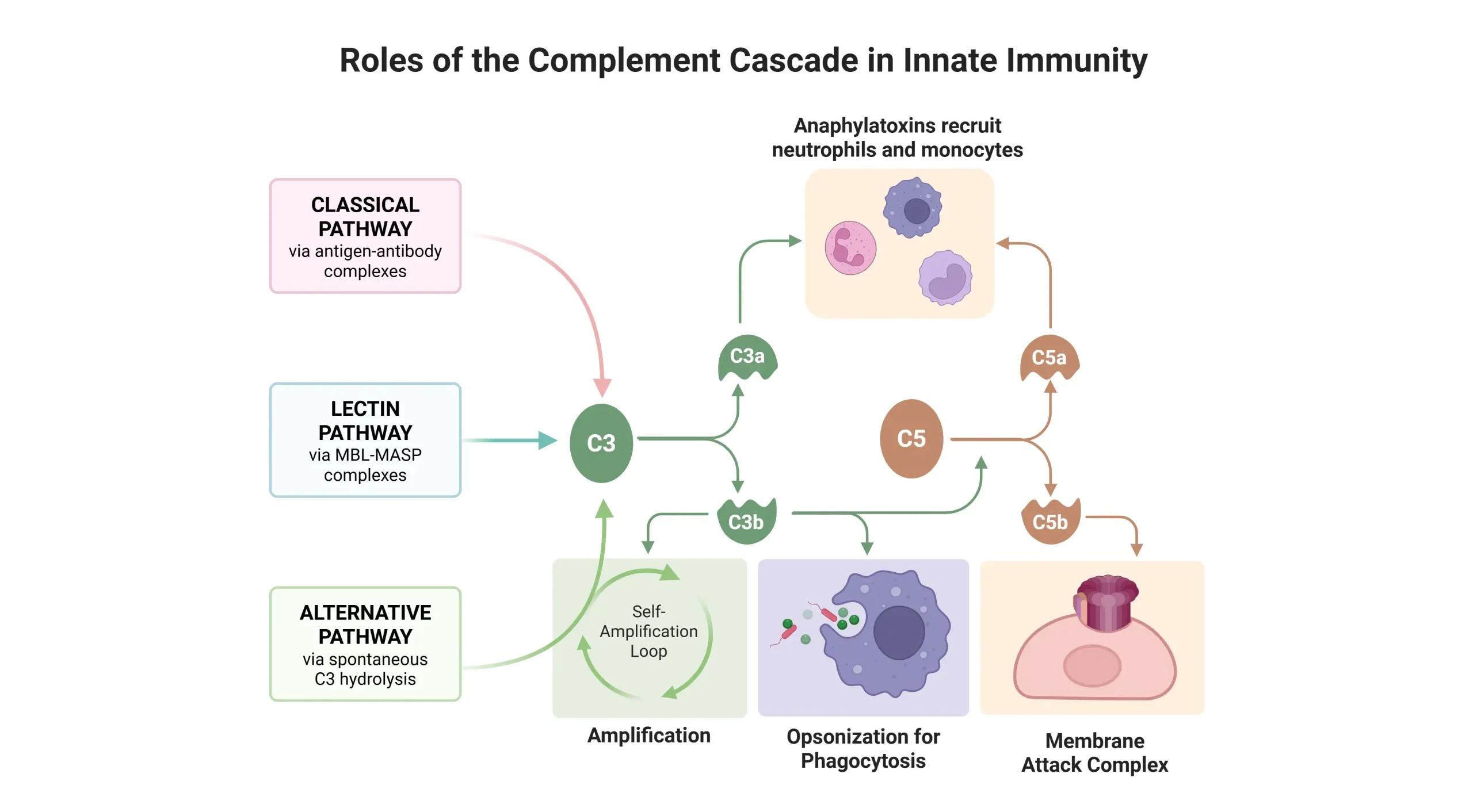
Created with BioRender.com
Activation Pathways
- Classic – IgG & IgM mediated → CH50 test → Screens for classical pathway defects.
- Alternative – Direct microbial
- Lectin – Mannose/sugar recognition
Key Functions
- C3b → Opsonization, immune complex clearance.
- C3a, C4a, C5a → Anaphylaxis (mast cell/basophil activation).
- C5a → Neutrophil chemotaxis.
- C5b-9 (MAC) → Cytolysis.
Clinical Correlations
➢ C3 deficiency → Recurrent pyogenic infections (Strep, Staph), immune complex diseases (SLE, GN).
➢ C5-C9 deficiency → Neisseria infections (meningitis, gonorrhea).
➢ CD55 (DAF) deficiency → Paroxysmal Nocturnal Hemoglobinuria (PNH).
➢ C1 esterase inhibitor deficiency → Hereditary angioedema (bradykinin-mediated, worsened by ACE inhibitors).
How it is tested:
→ A 35-year-old man presents with dark urine in the morning, anemia, and thrombosis in the hepatic vein (Budd-Chiari syndrome). Flow cytometry → Low CD55/CD59 on RBCs. Dx?
Answer: PNH
→ A 25-year-old woman with a history of systemic lupus erythematosus (SLE) presents with recurrent infections and symptoms of immune complex-mediated inflammation. Laboratory tests reveal low complement levels, particularly C3 and C4. What is the underlying problem in this patient?
Answer: Impaired ability to remove C3b-coated immune complexes
→ A 15-year-old boy presents with recurrent episodes of painless swelling involving his face, lips, and extremities. He also reports occasional abdominal pain with no associated urticaria. His father has a similar history. Laboratory tests show low C4 levels and decreased C1 esterase inhibitor activity.
What is the most likely diagnosis?
Answer: Hereditary angioedema (due to C1 esterase inhibitor deficiency)
D/Dx:
Hereditary angioedema (HAE) causes recurrent, non-pruritic swelling due to C1 esterase inhibitor deficiency and bradykinin excess. Unlike anaphylaxis, it lacks urticaria, is not IgE-mediated, and does not respond to epinephrine or antihistamines. Treatment includes C1 inhibitor replacement or bradykinin inhibitors (e.g., icatibant).
Clinically Relevant Cytokines
| Cytokine | Produced by | Function | Clinical Relevance |
|---|---|---|---|
| IL-1 & IL-6 | Macrophages | Fever, acute inflammation, acute-phase protein production | Sepsis (fever, shock), inflammatory diseases (RA, COVID-19 cytokine storm) |
| TNF-α | Macrophages | WBC recruitment, vascular leak, cachexia | Septic shock, cachexia in malignancy, granuloma maintenance in TB |
| IL-8 | Macrophages | Neutrophil chemotaxis | “Clean up on IL-8” → Pus formation, infections |
| IL-12 & IFN-γ | Macrophages, Th1, NK cells | Macrophage activation, granuloma formation | Deficiency → Susceptibility to TB & mycobacterial infections |
| IL-2 | T cells | Stimulates T & NK cell growth | Used in immunotherapy (melanoma, RCC) |
| IL-4 & IL-5 | Th2 cells | IgE & IgG class switching (IL-4), IgA & eosinophil activation (IL-5) | Asthma, allergies, eosinophilia in parasitic infections |
| IL-10 | Th2 & Tregs | Anti-inflammatory, suppresses Th1 response | Overexpression → Increased susceptibility to chronic infections |
✔ IL-10, IL- 4, TGF-β → Anti-inflammatory → Attenuates immune response
✔ IL-1, IL-6, TNF-alpha → Acute inflammation
How it is tested:
A 45-year-old man from Guatemala presents with a chronic cough, hemoptysis, and weight loss. Chest X-ray shows upper lobe cavitations, and sputum is positive for acid-fast bacilli. Genetic mutation impairs Th1 differentiation, leading to disseminated disease. Which cytokine is deficient due to this mutation?
Answer: IL-12
IL-12 from macrophages induces Th1 differentiation → Th1 cells release IFN-γ → activates macrophages → granuloma formation
◆ Interferons Inhibits viral replication→ ↑ MHC-1 expression → ↑ killing by NK and CD-8 + T-cells
✔ Activates antitumor immunity
Cell surface proteins
| Cell Type | Key Markers |
|---|---|
| T Cells | CD3, CD28 (TCR signaling, co-stim) |
| Helper T | CD4, CD40L (HIV, B-cell activation) |
| Regulatory T | CD4, CD25 (Immune suppression) |
| B Cells | CD19, CD20, CD21 (EBV), CD40 (T-cell interaction) |
| Macrophages | CD14 (LPS receptor), CD40, MHC II, Fc/C3b (phagocytosis) |
| NK Cells | CD16 (ADCC), CD56 (NK marker) |
| Stem Cells | CD34 (Hematopoiesis) |
High Yield Vaccines
| Vaccine Type | Key Features | High-Yield Examples |
|---|---|---|
| Live Attenuated | Strong cellular & humoral response. Long-lasting, but avoid in pregnancy & immunodeficiency due to reversion risk. | MMR, Varicella, Oral polio, Yellow fever “Live! MVSY” (MMR, Varicella, Sabin, Yellow fever) |
| Killed/Inactivated | Only humoral response. Needs boosters, safer than live. | Rabies, Hep A, Influenza (injected), Salk polio (Inj) “RIP Always” (Rabies, Influenza, Polio [Salk], Hep A) |
| Subunit | Only antigenic parts are used. Weaker response. | HBV (HBsAg), HPV, Meningococcus, Pneumococcus, Pertussis |
| Toxoid | Inactivates bacterial toxins, provides strong antibody-mediated immunity, but requires boosters. | “Toxic T&D” Tetanus, Diphtheria |
➢ Egg allergy🥚❌ → Avoid Yellow fever, Intranasal influenza (live)
➢ Live vaccines🚫 Contraindicated in:
- Pregnancy🤰
- Immunodeficiency(e.g., X-linked agammaglobulinemia, SCID)
- HIVwith CD4 < 200
➢ Polysaccharide vaccines → Only advantage ✅ ↓ Replacement strains (due to lack of mucosal immunity)
How it is tested:
→ A 34-year-old man with HIV presents for routine follow-up. He has been on antiretroviral therapy with good adherence. His CD4 count is 300 cells/mm³, and his viral load is undetectable. He is due for several vaccinations, including MMR and varicella. What you advise to the patient?
Answer: Live attenuated vaccines (MMR, Varicella) can be given safely to HIV patients if CD4 count ≥ 200 cells/mm³.
→ A 3-year-old boy arrives for routine vaccination. His parents, recently emigrated from a polio-endemic region, ask whether the oral polio vaccine (OPV, Sabin) or inactivated polio vaccine (IPV, Salk) provides better protection. Which is the most appropriate recommendation?
Answer: OPV (Sabin, live) → Stronger immunity (IgG + IgA) → Prevents transmission → give herd immunity
IPV (Salk, killed) → Weaker mucosal immunity (IgG only)
Hypersensitivity types
| Type | Mechanism | Key Mediators | Examples |
|---|---|---|---|
| Type I (Immediate, IgE) | IgE → Mast cell degranulation → Histamine, Leukotrienes | Histamine, Tryptase (marker), LTC₄, LTD₄, LTE₄ | Anaphylaxis, Allergic asthma |
| Type II (Antibody-mediated, IgG/IgM) | Antibodies bind cell antigens → Destruction, Inflammation, Dysfunction | IgG, IgM, C3b (opsonization), C5a (inflammation) | Destruction: AIHA, ITP, Transfusion reaction Inflammation: Goodpasture, Rheumatic fever Dysfunction: Myasthenia gravis, Graves |
| Type III (Immune Complex, IgG) | Immune complexes deposit → Complement activation → Neutrophil damage | Immune complexes, ↓C3, ↓C4 | SLE, RA, PSGN, Serum sickness-like reactions from some drugs such as penicillins and monoclonal antibodies |
| Type IV (Delayed, T-cell mediated) | CD8+ T cells: Direct killing CD4+ T cells: IFN-γ → Macrophage activation → TNF-α | T cells, IFN-γ, TNF-α | Contact dermatitis, TB test (PPD), GVHD, Multiple Sclerosis |
How it is tested:
→ A 25-year-old man with seasonal allergies develops sudden SOB, wheezing, and hypotension after eating peanuts. Exam shows urticaria and facial swelling. Symptoms improve with epinephrine. Which test best confirms the underlying diagnosis?
Answer: Serum tryptase → Most specific marker of mast cell activation in anaphylaxis.
→ A 28-year-old woman with weight loss, palpitations, and exophthalmos has low TSH, high free T4, and thyroid-stimulating immunoglobulins. Which type of hypersensitivity is responsible?
Answer: Type II
→ A 25-year-old hiker develops an intensely pruritic, erythematous rash with vesicles in a linear pattern on his forearms two days after hiking through a wooded area. Cytokine primarily responsible for this presentation?
Answer: IFN-γ (produced by Th1 cells, activates macrophages, and mediates tissue damage in Type IV hypersensitivity). It’s a contact dermatitis due to exposure to poison ivy, present in many plants
USMLE Hack:
- Type II: → Localized damage (1–2 organs; e.g., RBCs in AIHA, neuromuscular junction in MG).
- Type III: → Systemic involvement (≥3 organs; e.g., SLE, serum sickness).
Transfusion Reactions
| Reaction Type | Cause | Key clinical feature |
|---|---|---|
| Allergic/Anaphylactic Reaction → Minutes to 2-3 hrs | Type I hypersensitivity to plasma proteins (IgA in IgA-deficient) | Urticaria, pruritus, wheezing, hypotension, shock |
| Acute Hemolytic Transfusion Reaction → During transfusion or within 24 hrs | Type II hypersensitivity (ABO incompatibility) Mainly by IgM, also IgG | Fever, hypotension, hemoglobinuria, jaundice, flank pain. |
| Febrile Non-Hemolytic Reaction → 1-6 hrs | Cytokines from donor WBCs | Fever, chills, flushing, headache |
| Transfusion-Related Acute Lung Injury (TRALI) → Minutes to 6 hrs | Recipient Neutrophil against donor antibodies | Respiratory distress, non-cardiogenic pulmonary edema |
| Delayed Hemolytic Reaction → Onset over 24 hrs, usually 1-2 weeks | Anamnestic response to minor RBC antigens | Mild fever, hyperbilirubinemia, clinically silent |
How it is tested:
A 26-year-old woman with iron deficiency anemia develops chills, headache, and a mild fever (38.1°C) 2 hours after a blood transfusion. Vitals are stable, and there are no signs of hemolysis or respiratory distress. What is the most likely cause?
Answer: Cytokines accumulated in stored donor leukocytes (Febrile Non-Hemolytic Transfusion Reaction).
Immunosuppressants
| Drug | MOA | Unique Toxicities |
|---|---|---|
| 1. Cyclosporine | Calcineurin inhibitor → ↓ IL-2 transcription → ↓ T-cell activation | Nephrotoxicity, gingival hyperplasia, hirsutism |
| 2. Tacrolimus (FK506) | Same as cyclosporine but binds FKBP → ↓ IL-2 transcription | Nephrotoxicity, ↑ risk of diabetes |
| 3. Sirolimus (Rapamycin) DOC for kidney transplant prophylaxis | mTOR inhibitor → Blocks T & B-cell activation by ↓ response to IL-2 | Pancytopenia, insulin resistance, hyperlipidemia, NOT nephrotoxic |
| 4. Basiliximab | Monoclonal antibody → Blocks IL-2R | Tremor |
| 5. Azathioprine | 6-MP prodrug → Inhibits nucleotide synthesis → ↓ lymphocyte proliferation | Pancytopenia (Toxicity ↑ with allopurinol due to XO metabolism) |
| 6. Mycophenolate Mofetil | Inhibits IMP dehydrogenase → ↓ purine synthesis in B & T cells | CMV infection risk |
| 7. Glucocorticoids | Inhibit NF-κB → Suppress B & T cells + Induce T-cell apoptosis | Osteoporosis, Avascular necrosis |
How it is tested:
A 52-year-old man with end-stage renal disease undergoes a kidney transplant and is started on immunosuppressive therapy. Two weeks later, he developed hypertension, hyperlipidemia, and mild pancytopenia. His creatinine remains stable. Immunosuppressant likely responsible for his current findings?
Answer: Sirolimus → used for kidney transplant prophylaxis → NOT nephrotoxic
Immunodeficiency Disorders
| Disorder | Pathogenesis | Clinical Features | Key Findings |
|---|---|---|---|
| B-Cell Disorders | |||
| X-linked (Bruton) Agammaglobulinemia (XR) | BTK mutation → No B-cell maturation | Recurrent bacterial, Giardia & enteroviral infections after 6 months | ↓ B cells, ↓ IgG, IgA, IgM, Absent lymph nodes/tonsils |
| Selective IgA Deficiency | Defective IgA production | Mostly asymptomatic; ↑ risk of GI, airway infections, anaphylaxis to blood products | ↓ IgA, Normal IgG/IgM, False-negative celiac test |
| T-Cell Disorders | |||
| DiGeorge Syndrome (22q11) | 3rd & 4th pharyngeal pouch defect → Absent thymus & parathyroids | Viral/fungal infections, tetany (hypocalcemia), TOF, TGA | ↓ T cells, ↓ PTH, ↓ Ca²⁺, Absent thymic shadow |
| Hyper-IgE Syndrome (Job Syndrome, AD) | STAT3 mutation → Impaired Th17 function → ↓ Neutrophil recruitment | Cold staph abscesses, coarse facies, retained primary teeth, fractures, eczema | ↑ IgE, ↑ eosinophils |
| Combined B- and T-Cell Disorders | |||
| Severe Combined Immunodeficiency (SCID) (XR/AR) | IL-2Rγ mutation (XR) or ADA deficiency (AR) → No T/B cells | Failure to thrive, chronic diarrhea, recurrent infections (all types) | ↓ TRECs, Absent thymic shadow, germinal centers, T-cells |
| Hyper-IgM Syndrome (XR) | CD40L defect → No class switching | Severe pyogenic infections, PCP, CMV, Cryptosporidium | ↓ IgG, IgA, IgE, Normal IgM |
| Phagocyte Disorders | |||
| Leukocyte Adhesion Deficiency (LAD-1) (AR) | CD18 defect (LFA-1 integrin) → Impaired neutrophil migration | Delayed umbilical cord separation, recurrent non-purulent infections | Marked neutrophilia |
| Chronic Granulomatous Disease (CGD) (XR) | NADPH oxidase deficiency → ↓ ROS & respiratory burst | Recurrent skin abscesses, osteomyelitis, granulomas | Catalase+ organisms (S. aureus, Serratia, Nocardia, Aspergillus), DHR test: ↓ green fluorescence |
| Chediak-Higashi Syndrome (AR) | LYST mutation → Defective lysosomal trafficking | Albinism, recurrent pyogenic infections, neuropathy | Giant granules in neutrophils, pancytopenia |
How it is tested:
→ A 28-year-old man with recurrent respiratory & GI infections develops wheezing, hypotension, and respiratory distress minutes after a blood transfusion. Symptoms are resolved with epinephrine. Labs show undetectable IgA with normal IgG & IgM. Diagnosis?
Answer: Selective IgA Deficiency
→ Recommendations for future blood transfusion in this patient?
Answer: Washed RBCs or IgA-deficient donor blood to prevent anaphylaxis. Avoid plasma-containing products.
How it is tested:
A 3-day-old male neonate presents with poor feeding, stridor, and intermittent tetanic spasms. Exam reveals low-set ears, cleft palate, and a weak cry. Labs show hypocalcemia, low PTH, and lymphopenia. A flow cytometry test confirms a low CD3+ T-cell count. Complications this patient is at greatest risk for?
A) Pneumocystis jirovecii pneumonia
B) Autoimmune hemolytic anemia
C) Meningococcal sepsis
D) Anaphylaxis from blood transfusion
Answer: A) Pneumocystis jirovecii pneumonia → DiGeorge Syndrome
How it is tested:
→ A 2-month-old boy with failure to thrive, chronic diarrhea, recurrent infections (oral thrush, Pneumocystis), absent tonsils, and an absent thymic shadow on CXR. Labs show profound lymphopenia. Which is the best initial test?
Answer: SCID → T-cell receptor excision circles (TRECs)
How it is tested:
→ A 7-year-old boy has recurrent skin and lung infections with Staphylococcus aureus and Aspergillus. He develops granulomatous lesions at infection sites. Dihydrorhodamine (DHR) flow cytometry shows decreased green fluorescence. Which of the following is the underlying defect?
Answer: CGD → NADPH oxidase deficiency
→ A 5-year-old boy is hospitalized for recurrent pneumonia and skin abscesses. Cultures repeatedly grow Serratia marcescens and Burkholderia cepacia. His neutrophils fail to produce a respiratory burst on dihydrorhodamine (DHR) flow cytometry testing. Which additional pathogens is he at greatest risk for?
Answer: Patients with chronic granulomatous disease (CGD) are at increased risk for infections caused by catalase-positive organisms. The most high-yield pathogens include:
- Staphylococcus aureus
- Burkholderia cepacian
- Serratia marcescens
- Nocardia
- Aspergillus
Transplant Rejection
| Type | Cause | Immune Mechanism | Clinical Features |
|---|---|---|---|
| Hyperacute (Minutes-during surgery) | Pre-existing antibodies (Type II HSR) Due to anti-ABO or anti-HLA antibodies | Antibody-mediated, complement activation | Widespread thrombosis, ischemia, necrosis; AKA ‘white graft rejection’ Graft removal required |
| Acute (Weeks–Months) | Donor MHC activates T & B cells | Cellular: CD8+ & CD4+ T cells (Type IV HSR) Humoral: Antibodies (C4d deposition) | Vasculitis with lymphocytic infiltrate; Reversible with immunosuppression |
| Chronic (Months–Years) | CD4+ T cells respond to donor peptides Vascular wall thickening and luminal narrowing | Cellular & Humoral (Type II & IV HSR) | Arteriosclerosis, fibrosis, parenchymal atrophy, organ-specific failure (e.g., chronic allograft nephropathy, bronchiolitis obliterans)—Refractory to immunosuppressants |
How it is tested:
→ A 55-year-old man with end-stage renal disease undergoes a kidney transplant. Minutes after the vascular clamps are released, the transplanted kidney becomes mottled and cyanotic. The surgeon notes poor urine output and immediate graft dysfunction. What is the immune mechanism responsible for this patient’s condition?
Answer: Hyperacute rejection → anti-ABO or anti-HLA antibodies
→ A 35-year-old man undergoes a kidney transplant. Ten weeks later, he presented with fever, reduced urine output, and rising creatinine levels. Physical examination reveals tenderness over the graft site. Doppler ultrasound shows increased vascular resistance in the transplanted kidney. Which of the following findings are most likely on biopsy?
Answer: Interstitial lymphocytic infiltrate
If you need guidance on your target USMLE scores or have questions on improving your chances of matching into your dream specialty, feel free to schedule a call here or text us on WhatsApp here for personalized support.
🍀 We wish you best of luck on your exam!


2026 Residency Personal Statement The Ultimate Guide to Crafting Your Residency Personal Statement

Best Resources for USMLE STEP 1! How to Study and Prepare for STEP1?

ERAS Application 2026 Guide! How to Fill Out ERAS Residency Application?
About TheMatchGuy
RECOMMENDED ARTICLES

Best Electives in the US

Best Resources for USMLE STEP 1! How to Study and Prepare for STEP1?

United States Clinical Experience for IMGs