USMLE Blog
USMLE Step 1 High‐Yield Pharmacology Topics


If you want our team’s expert help with your USMLE prep, click here
Pharmacology
High Yield USMLE Step 1 Notes
Welcome to the Pharmacology High-Yield Notes for Step 1.
These are the most commonly tested topics on the exam—ideal for quick review in your last 2 weeks before the exam, when you’re short on time, or when you’re just starting out and want to focus on what is high yield. Created and peer-reviewed by physicians who scored 260+ on Step 1 in a Q&A style to reinforce active learning.
How to use this file
- Read the question ➔ cover the answer ➔ self-quiz before revealing.
- Review the NOTES ➔ they include high-yield details you don’t want to miss.
- Need tutoring? The Match Guy USMLE Tutoring.
- Don’t forget to watch the video lessons that go with this file. Learn more here.
Table of Contents
Pharmacokinetics & Pharmacodynamics
1. A 60M with alcoholic cirrhosis presents with confusion and signs of drug toxicity. He is taking a highly protein-bound antiarrhythmic medication. How does his liver disease affect the drug’s Vd?
➔ Increased
- Note: Volume of distribution Vd is increased due to lower plasma protein binding.
- Liver disease reduces albumin, increasing free drug levels and expanding Vd.
- More drug in tissues reduces the efficacy of dialysis for removal.
2. A 70M with CHF and significant peripheral edema is started on a hydrophilic diuretic. How does fluid overload affect the drug dosing and volume of distribution?
➔ Fluid overload increases the volume of distribution, potentially requiring a higher loading dose to achieve therapeutic plasma levels
- Note: Hydrophilic drugs stay mainly in extracellular fluid, which is expanded in edema and CHF.
- Increased Vd dilutes the drug concentration in plasma, reducing initial effectiveness if dose isn’t adjusted.
| Mnemonic “PET” | Drug Characteristics | Dialysis Effectiveness |
|---|---|---|
| P | Large, charged, protein-bound → Remains in Plasma → low Vd | High (easily dialyzed) |
| E | Small, hydrophilic → Stays mainly in ECF → Medium Vd | Moderate |
| T | Small, lipophilic → Distributes into Tissues (incl. fat) → High Vd | Low (poorly dialyzed) |
3. A patient is prescribed a new medication that is eliminated at a constant rate regardless of plasma concentration. After increasing the dose, the patient developed signs of toxicity. Which elimination kinetics does this drug follow?
➔ Zero-order elimination
- Note: Zero-order drugs eliminate at a constant rate or amount per unit time due to enzyme saturation.
- Plasma concentration decreases linearly, not exponentially.
- Examples: Phenytoin, ethanol, aspirin (at toxic doses).
- First-order drugs: constant proportion or fraction of drug eliminated per unit time.
4. A 45-year-old man overdoses on aspirin and develops toxicity. Why does aspirin elimination shift from first-order to zero-order kinetics at high doses?
➔ Enzyme saturation causes elimination to proceed at a constant rate, independent of plasma concentration
- Note: At therapeutic doses, aspirin elimination is first-order.
- At toxic doses, elimination enzymes saturate, leading to zero-order kinetics and risk of accumulation.
Key Difference:
✓ Zero-order: Elimination rate is constant (enzyme saturation).
✓ First-order: Elimination rate varies with concentration (proportional elimination).
5. A factory worker collapses with lactic acidosis and cherry-red skin after inhaling fumes. Increasing oxygen fails to improve symptoms. What explains this?
➔ Cyanide causes noncompetitive inhibition of cytochrome oxidase (Complex IV), decreasing Vmax irreversibly
- Note: Noncompetitive inhibitors bind allosteric sites → ↓ Vmax, no change in Km.
- Increasing substrate (O₂) does not overcome inhibition.
6. A 45-year-old man ingests antifreeze and develops metabolic acidosis. He is treated with IV ethanol. What is ethanol’s mechanism of action?
➔ Ethanol is a competitive inhibitor of alcohol dehydrogenase, preventing formation of toxic metabolites from methanol
- Note: Competitive inhibitors resemble substrates and increase Km but do not affect Vmax.
- Increasing substrate concentration can overcome competitive inhibition.
| Feature | Competitive Inhibitors | Noncompetitive Inhibitors |
|---|---|---|
| Structure | Resembles substrate | Binds allosteric site |
| Overcome by ↑ [S] | Yes (e.g., statin vs. HMG-CoA) | No |
| Effect on Km | ↑ Km (↓ affinity) | No change |
| Effect on Vmax | No change (reversible) | ↓ Vmax (e.g., lead poisoning) |
7. A heroin-dependent patient develops sudden withdrawal after starting buprenorphine. What explains this reaction?
➔ Buprenorphine displaces heroin but provides weaker μ-receptor activation
- Note: Buprenorphine = partial μ-opioid agonist → high affinity, low efficacy.
- Displaces full agonists (e.g., heroin) from receptors.
- Weaker activation → precipitated withdrawal in opioid-dependent patients.
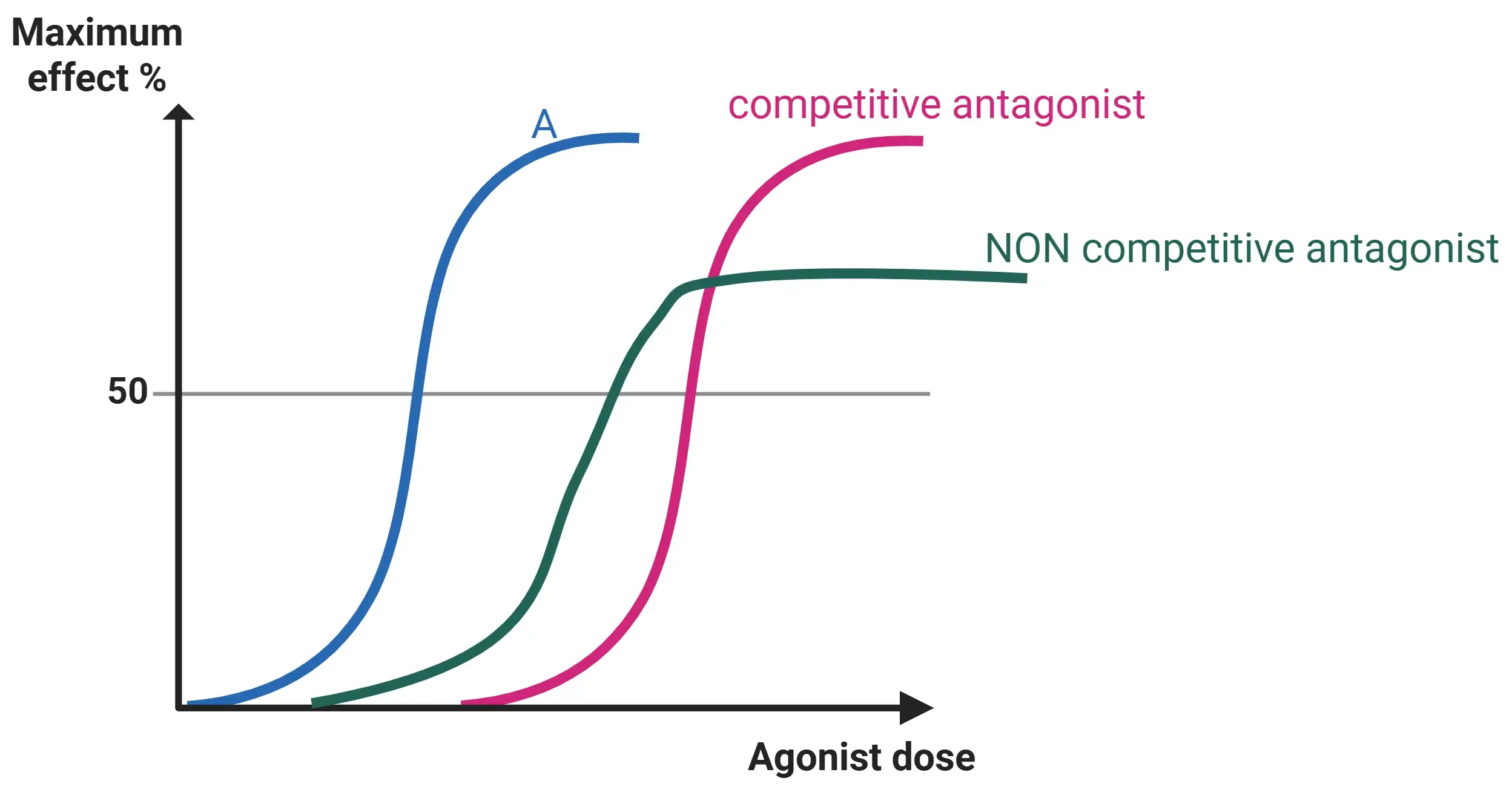
8. In dose-response testing of two antihypertensive drugs: Drug X requires a low dose but has a limited maximal effect; Drug Y requires a higher dose but achieves greater maximal effect. Which drug is more potent and which is more efficacious?
➔ Drug X is more potent (lower dose needed), Drug Y is more efficacious (higher maximal effect)
- Note: Potency = dose needed for effect (related to affinity).
- Efficacy = maximum effect achievable, independent of potency.
Urine pH & drug elimination
9. A 24-year-old woman presents to the ED after ingesting a large amount of aspirin. IV sodium bicarbonate is administered. Primary mechanism of this intervention?
➔ Alkalinization of urine
- Note: Aspirin is a weak acid.
- Alkaline urine increases ionization, preventing reabsorption and enhancing excretion.
10. A patient with amphetamine toxicity is given ammonium chloride. Rationale for this intervention?
➔ Acidic urine increases ionization of amphetamines, preventing reabsorption
- Note: Weak bases like amphetamines are better cleared in acidic urine.
- Ionized form remains trapped in renal filtrate.
11. You are treating a patient who ingested an unknown substance with a pKa of 8.2. At physiological pH (7.4), would the drug predominantly be ionized or non-ionized?
➔ Predominantly ionized
- Note: Weak bases are ionized when PH <pka → ↓ absorption & reabsorption.
- Non-ionized form crosses membranes easily → ↓ renal clearance.
- Urine acidification traps the drug in ionized form → ↑ excretion.
General anesthetics
12. A patient under general anesthesia with an inhaled agent develops severe muscle rigidity, hypercarbia, tachycardia, and hyperthermia. Anesthetic likely responsible?
➔ Isoflurane (or any volatile anesthetic except nitrous oxide)
- Note: Malignant Hyperthermia:
- Due to excessive calcium release from the sarcoplasmic reticulum → sustained contraction, hypermetabolism.
- Presents with muscle rigidity, hypercarbia, tachycardia, hyperthermia, acidosis, rhabdomyolysis.
- Triggering Agents: Volatile anesthetics (isoflurane, sevoflurane, desflurane) and succinylcholine.
- Treatment: Dantrolene (ryanodine receptor antagonist).
- Halothane can cause hepatotoxicity. use Sevoflurane in those with liver diseases.
13. A patient requires rapid induction and recovery during surgery. Which inhaled anesthetic is the best choice?
➔ Desflurane or Nitrous Oxide (lowest blood solubility → fastest induction/recovery)
- Note: Desflurane has low potency but very low blood solubility.
- Nitrous oxide has the lowest potency and fastest onset.
| Inhaled Anesthetic | Lipid Solubility (Potency – MAC↓) | Blood Solubility (Induction/Recovery) |
|---|---|---|
| Halothane | High (Very potent) | High (Slow induction/recovery) |
| Nitrous Oxide | Very Low (Least potent) | Very Low (Fastest induction) |
📝 Trick to remember:
- “High Lipid = High Potency = Low MAC”
- “High Blood = Slow Induction”
Maintenance & Loading Dose
14. A 65F with Hx of liver cirrhosis is prescribed warfarin, which is primarily metabolized by the liver. Later, she presents with elevated INR levels. What pharmacokinetic change is responsible for this?
➔ Liver cirrhosis → ↓ hepatic clearance → ↑ half-life → drug accumulation → increased bleeding risk
- Note: Clearance (CL): Rate at which a drug is eliminated from the body.
- Liver Cirrhosis Impact: Reduced metabolism → prolonged drug action → toxicity.
- Clinical Implication: Lower doses or longer intervals may be required to prevent toxicity.
15. A 68M with sepsis is started on vancomycin. His physician notes an increased volume of distribution (Vd) due to his body habitus. How should the loading dose be adjusted?
➔ Increased Vd → drug distributes more widely → higher loading dose needed to achieve therapeutic levels
- Note: Volume of Distribution (Vd): Indicates how extensively a drug spreads into body tissues.
- Obesity → more tissue mass → greater distribution → higher loading dose required.
- Clinical Implication: Dosing must account for Vd to reach effective plasma concentration.
Key takeaway:
✓ ↑ Vd → ↑ t₁/₂
✓ ↑ CL → ↓ t₁/₂
Impaired liver or kidney function → ↓ clearance → ↑ half-life → prolonged drug exposure → increased toxicity risk.
16. A 72-year-old woman with chronic kidney disease is started on digoxin. A week later, she presents with nausea, visual disturbances, and bradycardia. What explains the toxicity?
➔ Renal impairment → ↓ clearance → ↑ half-life → digoxin accumulation → toxicity
- Note: In patients with renal impairment, the dose of digoxin must be reduced, and serum levels should be monitored to prevent toxicity.
- Management: Monitor renal function regularly, adjust dose based on GFR, and consider digoxin-specific antibody (Digibind) if toxicity is severe.
- Renal dysfunction reduced insulin and morphine metabolite clearance, causing hypoglycemia and sedation, requiring dose reduction.
- Adjust the loading dose based on Vd and the maintenance dose based on CL to ensure proper drug levels and avoid toxicity.
Drug & enzyme reactions
17. A 60-year-old patient with atrial fibrillation is started on warfarin. He is also taking rifampin. How would this affect his maintenance dose?
➔ Rifampin → increased clearance of warfarin → higher maintenance dose required
- Note: Enzyme Induction: Rifampin accelerates warfarin metabolism.
- Monitor INR frequently and adjust the dose.
18. A 72F with generalized anxiety is prescribed diazepam. She experiences confusion and excessive sedation. Which phase of drug metabolism is most likely impaired in elderly patients?
➔ Phase I (Oxidation)
- Note: Phase I Reactions: Oxidation, Reduction, Hydrolysis (Old Rats Hide)
- Elderly lose Phase I first → “Old People Fade First”
19. A patient on warfarin is started on ciprofloxacin for a UTI. One week later, her INR is significantly elevated. Which drug interaction is responsible?
➔ CYP450 Inhibition (Ciprofloxacin)
- Note: CYP450 Inhibitors: SICKFACES.COM
- Effect: ↓ Warfarin breakdown → ↑ INR and bleeding risk
| CYP-450 Inhibitors (“SICKFACES.COM”) | Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole |
| CYP-450 Inducers (“CRAP GPS”) | Carbamazepine, Rifampin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, St. John’s Wort |
20. A 45M with COPD on theophylline presents with vomiting, insomnia, tremors, palpitations. He recently started ciprofloxacin for a UTI. Likely cause of his Sx?
➔ Theophylline toxicity due to CYP450 inhibition by ciprofloxacin
- Note: Theophylline has a narrow therapeutic index — small changes in dose or metabolism can lead to toxicity.
- CYP1A2 inhibitors (e.g., ciprofloxacin, cimetidine, erythromycin, verapamil) reduce metabolism → toxic accumulation.
- Toxicity signs:
○ GI: nausea, vomiting
○ CNS: tremors, insomnia, seizures
○ CV: palpitations, tachyarrhythmias
21. A 55-year-old man on isoniazid therapy tests positive for anti-histone antibodies. What genetic trait is responsible?
➔ Slow Acetylation
- Note: SHIP Drugs: Sulfasalazine, Hydralazine, Isoniazid, Procainamide → Drug-induced lupus
- Mechanism: Slow metabolism → drug accumulation
Key takeaway:
✔ Slow acetylators → More drug accumulation → Higher toxicity risk
✔ Fast acetylators → Rapid clearance → Lower drug efficacy
Sympathetic and Parasympathetic
22. A 30M presents with excessive sweating and is diagnosed with primary hyperhidrosis. He is prescribed a topical drug. Which neurotransmitter is blocked by the drug to reduce sweating?
➔ Acetylcholine
- Note: Sweat glands are sympathetic but use ACh (M₃), not NE..
- Blocking M₃ reduces sweating.
Key takeaway:
✔ Sweat glands = Sympathetic but use ACh instead of norepinephrine.
✔ Adrenal medulla = Direct preganglionic innervation
23. A 72M with BPH is prescribed tamsulosin. A week later, he reports dizziness when standing. MOA of this side effect?
➔ Blocks α₁ receptors → vasodilation → orthostatic hypotension
- Note: α₁ blockers (tamsulosin) relax prostate smooth muscle but may cause postural hypotension by blocking α₁ receptors in vessels.
24. A patient with asthma uses albuterol during an acute episode and gets better. Which receptor is stimulated?
➔ β₂ agonist → bronchodilation
- Note: β₂ activation → ↑cAMP → smooth muscle relaxation → bronchodilation.
- However, at high doses, β₁ receptors in the heart can be secondarily stimulated, causing tachycardia and palpitations.
- Avoid in patients with CAD.
25. A 30-year-old man develops sudden-onset wheezing, hypotension, and urticaria after a bee sting. What is DOC?
➔ IM epinephrine
- Note: Epinephrine (α ≈ β)
- α₁ → vasoconstriction → ↑ BP, ↓ mucosal edema
- β₁ → ↑ HR & CO
- β₂ → bronchodilation
- Rapidly reverses airway obstruction and hypotension → first-line treatment for anaphylaxis.
26. A 43M presents with distended abdomen after inguinal hernia repair. USG shows post-void retention. Which muscarinic agonist would be most appropriate?
➔ Bethanechol
- Note: M₃ agonist → activates detrusor muscle → promotes urination.
- Used for post-op or neurogenic bladder.
- blocking M₃ by oxybutynin= useful for urinary urgency and incontinence.
27. A 38-year-old woman exposed to insecticides develops miosis, bradycardia, and salivation. DOC?
➔Atropine
- Note: Organophosphate Poisoning
- Cause: Inhibition of acetylcholinesterase → Excess acetylcholine → Cholinergic toxicity
- Some mushrooms produce similar effects.
- Clinical Features (DUMBBELLS): Diarrhea, Urination, Miosis, Bradycardia, Bronchospasm, Bronchorrhea, Emesis, Lacrimation, Lethargy, Salivation, Sweating
- Treatment:
1. Atropine (muscarinic antagonist)
2. Pralidoxime (2-PAM) (reactivates acetylcholinesterase)
28. A 50-year-old man with CKD presents with hypertensive emergency. He is given IV fenoldopam. MOA?
➔ D₁ receptor agonist → vasodilation of renal arteries → ↓BP
- Note: Fenoldopam is a short-acting D₁ agonist.
- Used in hypertensive emergencies, especially in patients with renal insufficiency.
- It promotes natriuresis and maintains renal blood flow
29. A 60-year-old man with GERD is prescribed famotidine. What is its target and mechanism?
➔ H₂ receptor antagonist → ↓gastric acid secretion
- Note: H₂ blockers (e.g., ranitidine, famotidine) are used to treat GERD & peptic ulcers by blocking histamine in parietal cells.
- H1- blockers like diphenhydramine are used for allergic rhinitis
30. A 65-year-old with Alzheimer’s disease is started on donepezil. What receptor is targeted and what is the effect?
➔ M₁ agonist (via cholinesterase inhibition) → enhances cognition
- Note: M₁ receptors in CNS help with memory.
- Donepezil is an AChE inhibitor used in Alzheimer’s.
31. A 62-year-old man with newly diagnosed CHF is started on dobutamine. Which receptor does this drug primarily act on and what is the effect?
➔ β₁ receptor agonist → ↑ heart rate and contractility
- Note: β₁ receptors are found in the heart and kidneys.
- Dobutamine = β₁ agonist → used in acute heart failure to increase cardiac output.
32. A 47-year-old woman is prescribed metoclopramide for nausea. What is the drug’s mechanism of action?
➔ D₂ receptor antagonist → blocks dopamine in the CTZ (chemoreceptor trigger zone)
- Note: D₂ blockade in the CNS → antiemetic effect.
- Commonly used in gastroparesis and chemo-induced nausea.
33. A 10-year-old boy is started on methylphenidate for ADHD. What is the mechanism of action?
➔ Reverses NET → ↑ NE and dopamine in the synapse → improves attention and focus
- Note: ADHD stimulants (methylphenidate, amphetamines) enhance catecholamine release by reversing NET and DAT. used in ADHD and Narcolepsy.
34. A 45-year-old woman on MAO inhibitors uses cocaine recreationally. She presents with severe hypertension, headache, and agitation. Why is her crisis worse?
➔ Cocaine blocks NE reuptake (↑ NE); MAOIs prevent NE breakdown → massive NE buildup → hypertensive crisis
- Note: Combining reuptake inhibitors (like cocaine/TCAs) with MAOIs causes catecholamine storm.
- Avoid tyramine-containing foods too (aged cheese).
35. A 70-year-old man is admitted with fever, dysuria and warm extremities, low BP (75/40), and elevated HR (110). What is the vasopressor of choice?
➔ Norepinephrine
- Note: Norepinephrine (α1 > β1) — increases MAP via vasoconstriction and maintains perfusion.
- NE is the DOC for septic shock. It increases both systolic and diastolic BP, leading to reflex bradycardia sometimes.
| Receptor | Location | Effect | Drugs |
|---|---|---|---|
| M₃ | Bladder detrusor, GI, glands, eye | SM contraction, ↑Secretions | 🟢Bethanechol → Urinary retention 🔴Oxybutynin → Urinary urgency |
| α₁ | Vascular SM, bladder sphincter | Vasoconstriction, urinary retention | 🔴Tamsulosin, prazosin → BPH |
| β₁ | Heart (SA/AV node), JG cells | ↑HR, ↑Contractility, ↑Renin | 🔴Metoprolol → HTN, arrhythmias 🟢Dobutamine → CHF |
| β₂ | Bronchial SM, uterus, skeletal vessels | Bronchodilation, SM relaxation | 🟢Albuterol → Asthma, preterm labor |
| D₂ | CNS (striatum, pituitary) | ↓Prolactin, ↓Nausea/Vomiting | 🔴Haloperidol → Psychosis 🟢Bromocriptine → Hyperprolactinemia |
| H₁ | Respiratory tract, skin | ↑Mucus, bronchoconstriction | 🔴Loratadine, Diphenhydramine → Allergic rhinitis, urticaria |
| H₂ | Gastric parietal cells | ↑Acid secretion | 🔴Famotidine → GERD, peptic ulcers |
| V₂ | Kidney (collecting duct) | ↑Water reabsorption via AQP2 | 🟢Desmopressin → Central DI, nocturnal enuresis 🔴Conivaptan → SIADH |
🟢:Agonist 🔴:Antagonist
36. A patient receives botulinum toxin for blepharospasm. What is the mechanism of action?
➔ Inhibits ACh release at NMJ → prevents muscle contraction
- Note: Botulinum toxin blocks presynaptic ACh release
- Used for muscle spasm, cosmetic use, and Diffuse Esophageal Spasm (DES)
37. Which drug class should be started first in a patient with pheochromocytoma preparing for surgery, and why is it is preferred over phentolamine?
➔ Phenoxybenzamine → irreversible α-blockade
- Note: Phenoxybenzamine = irreversible α-blocker
- Phentolamine = reversible α-blocker
- Starting β-blockers before α-blockade risks unopposed α stimulation → hypertensive crisis
Drug metabolism & elderly
38. A 78M on digoxin presents with nausea, visual changes, and confusion. Labs show impaired renal function and high digoxin levels. Why does toxicity develop despite normal dosing?
➔ ↓ Total body water in elderly → ↓ Vd for hydrophilic drugs like digoxin → drug accumulation
- Note: Elderly = ↓ renal clearance, ↓ total body water, altered distribution/metabolism.
- Renally cleared, narrow-TI drugs (digoxin, lithium) require dose adjustment.
39. A 60-year-old man with a history of peptic ulcer disease is started on ibuprofen for chronic back pain. What is the most important complication to consider?
➔ ↑ Risk of GI bleeding
- Note: NSAIDs inhibit prostaglandin synthesis → ↓ gastric mucus and bicarbonate → mucosal injury.
- Risk is higher in older adults and those with prior ulcers.
- Co-prescribe a PPI to reduce GI complications.
40. A 75F is given diphenhydramine for allergic rhinitis. She returns with confusion, dry mouth, and urinary retention. Why should diphenhydramine be avoided in older adults?
➔ Due to anticholinergic effects
- Note: Diphenhydramine = strong anticholinergic → ↑ risk of delirium, falls, urinary retention.
- Elderly have ↓ cholinergic reserve and slower clearance.
- Listed in Beers Criteria — avoid in older adults.
41. A 68-year-old man is taking multiple medications. His new drug is highly protein-bound. What happens if he has hypoalbuminemia?
➔ ↑ Free (active) drug concentration → ↑ toxicity risk
- Note: Low albumin (e.g., in liver disease, elderly) → less protein binding → more free drug → enhanced effect/toxicity of protein-bound drugs (e.g., warfarin, phenytoin).
42. A 74-year-old man started on diazepam for anxiety. What pharmacokinetic property makes this drug risky in the elderly?
➔ ↓ Phase I metabolism → prolonged half-life and sedation
- Note: Diazepam undergoes Phase I metabolism (oxidation), which declines with age.
- Safer options like lorazepam use Phase II (unchanged in elderly).
43. A 70-year-old woman on amitriptyline for neuropathic pain reports dizziness and near-syncope on standing. Vitals show orthostatic hypotension. What is the likely cause?
➔ α₁ receptor blockade by a tricyclic antidepressant (TCA)
- Note: TCAs (e.g., amitriptyline, nortriptyline) inhibit NET, block muscarinic, H₁, and α₁ receptors.
- α₁ blockade → vasodilation → orthostatic hypotension, especially in elderly.
- Other TCA side effects: anticholinergic toxicity, sedation, QT prolongation.
- Use caution in older adults.
Common drugs to avoid in elderly according to BEER’S criteria
- α-blockers: ↑ risk of hypotension
- Anticholinergics, TCAs, antihistamines, opioids: ↑ delirium, falls, constipation, urinary retention
- Benzodiazepines: ↑ sedation, falls, cognitive impairment
- NSAIDs: ↑ GI bleeding (esp. with anticoagulants)
- PPIs (long-term): ↑ risk of C. difficile infection
🏁 Final Thoughts
You made it to the end—well done! These high-yield notes are designed to save you time and keep you focused on what actually matters for Step 1. Review these vignettes regularly—steady practice beats cramming.
Need one-on-one tutoring? Visit The Match Guy USMLE Tutoring — we’ve helped hundreds of students reach their score goals.
Don’t forget to watch the video lessons that go with this file. They are part of our 7-day high-yield review of Step 1.
Join this RISK FREE live bootcamp 👉 Sign up here
Good luck and we’re rooting for you all the way to Match Day! 🎯
Disclaimer
Version 1.0 May 2025. No part of this document may be reproduced or distributed without written permission. For personal study only. Medical information herein is for educational purposes and does not constitute clinical advice. Always confirm with current guidelines and your institution’s policies.
Prefer a narrative review instead of Q&A style? Scroll down to the section below for high-yield concepts in paragraph format (they cover the same concepts).
Narrative Format
High-Yield Pharmacology Topics
High-yield general pharmacology for USMLE & NBME
Table of Contents
- Urine pH & Drug Elimination
- Volume of Distribution
- Elimination of Drugs
- Enzyme Kinetics & Inhibition
○ Enzyme Inhibition - Efficacy vs potency
- Clearance and Half-life
- Loading and Maintenance dose
- Drug metabolism
- Slow and Fast Acetylators
- Drug effect modifications
- Autonomic receptors
○ G Protein Coupled Receptors (GPCRs) - Adrenergic Receptors: Key Drug Targets
- Cholinergic Receptors: Key Drug Targets
- Dopamine Receptors: Key Drug Targets
- Histamine Receptors: Key Drug Targets
- Vasopressin Receptors: Key Drug Targets
- Drugs Acting in Autonomic Pathways
- Physiologic effects and uses of sympathomimetics
- Beers Criteria
- Age Related Pharmacokinetic Shifts
Urine pH & Drug Elimination
➔ ⬆️ Drug ionization = ⬆️ Trapping in urine = ⬆️ Excretion
➔ Neutral drug = ⬆️ Membrane passage = ⬆️ Reabsorption
Treatment of medication overdose:
➢ Weak Acids (e.g., aspirin, phenobarbital):
Alkaline urine (via sodium bicarbonate)
Weak Bases (e.g., TCAs, amphetamines):
Acidic urine (via vitamin C or ammonium chloride)
Key takeaway:
Sodium Bicarbonate in TCA Overdose (Exception): TCA is a weak base, but sodium bicarbonate is used to: – Alkalinize serum – Reduce TCA binding to cardiac sodium channels – Decrease arrhythmia risk (*Not for enhancing elimination*)
Rule of Thumb: At physiological pH (7.4), if the pKa is below 7.4, the drug behaves as a weak acid; if it’s above 7.4, it acts as a weak base.
How it is tested:
➔ A 25-year-old woman presents with tinnitus, nausea, and vomiting following an aspirin overdose. Labs reveal metabolic acidosis (pH 7.2). Knowing that aspirin is a weak acid (pKa <7.4), How do you treat it?
Answer: Alkalinize the urine by administering sodium bicarbonate.
Volume of Distribution
| Mnemonic “PET” | Drug Characteristics | Dialysis Effectiveness |
|---|---|---|
| P | Large, charged, protein-bound → Remains in Plasma → low Vd | High (easily dialyzed) |
| E | Small, hydrophilic → Stays mainly in ECF → Medium Vd | Moderate |
| T | Small, lipophilic → Distributes into Tissues (incl. fat) → High Vd | Low (poorly dialyzed) |
How it is tested:
➔ A 60-year-old man with a history of alcoholic cirrhosis is admitted with confusion and signs of drug toxicity. He is on a medication for arrhythmia that is highly protein-bound. What is the likely effect of his liver disease on the drug’s volume of distribution and its Dialyzability?
Answer: Increased Vd; decreased dialyzability
➔ A 70-year-old man with congestive heart failure and significant edema is started on a hydrophilic diuretic. Effect of fluid overload on the drug dosing?
Answer: Fluid overload (↑ ECF) → ↑ Vd for water-soluble drugs → Diluted plasma concentration → May require a higher dose to achieve therapeutic levels.
Elimination of Drugs
💊 Zero-Order Elimination (Constant Amount Lost)
➢ The same amount of drug is eliminated per unit of time (e.g., 2 mg/hr), regardless of concentration.
➢ Linear decrease in plasma concentration over time.
➢ Seen in PEA drugs: Phenytoin, Ethanol, Aspirin (at high/toxic doses).
➢ Why? Enzymes get saturated → can’t be eliminated faster even if more drugs are present.
💊 First-Order Elimination (Constant Fraction Lost)
➢ A fixed percentage of drugs is eliminated per unit time (e.g., 50% per hour).
➢ Exponential decrease in plasma concentration.
➢ Most drugs follow this pattern.
➢ Why? Elimination depends on drug concentration → higher concentration = faster elimination.
Key Difference:
✔ Zero-order: Elimination rate is constant (enzyme saturation).
✔ First-order: Elimination rate varies with concentration (proportional elimination).
How it is tested:
➔ A new drug is found to be eliminated at a constant rate, regardless of its plasma concentration. At higher doses, drug accumulation occurs, increasing the risk of toxicity. Which of the following best describes this drug’s elimination kinetics?
Answer: Zero-order elimination
Enzyme Kinetics & Inhibition
Enzyme Inhibition
| Feature | Competitive Inhibitors | Noncompetitive Inhibitors |
|---|---|---|
| Structure | Resembles substrate | Binds allosteric site |
| Overcome by ↑ [S] | Yes (e.g., statin vs. HMG-CoA) | No |
| Effect on Km | ↑ Km (↓ affinity) | No change |
| Effect on Vmax | No change (reversible) | ↓ Vmax (e.g., lead poisoning) |
How it is tested:
➔ A factory worker collapses after inhaling fumes. Exam reveals cherry-red skin, lactic acidosis, and ↓ O₂ saturation despite normal PaO₂. Why does increasing substrate (O₂) fail to reverse his symptoms?
Answer: Cyanide noncompetitively inhibits cytochrome oxidase (Complex IV) → irreversible ↓ Vmax of oxidative phosphorylation.
➔ A 45-year-old man presents with confusion, vomiting, and blurred vision after ingesting windshield washer fluid. Labs reveal a high anion gap metabolic acidosis and elevated osmolar gap. He is given intravenous ethanol as part of treatment. What is the MOA of ethanol in this scenario?
Answer: Ethanol competitively inhibits alcohol dehydrogenase, slowing the conversion of methanol to toxic metabolites (formic acid).
Efficacy vs potency

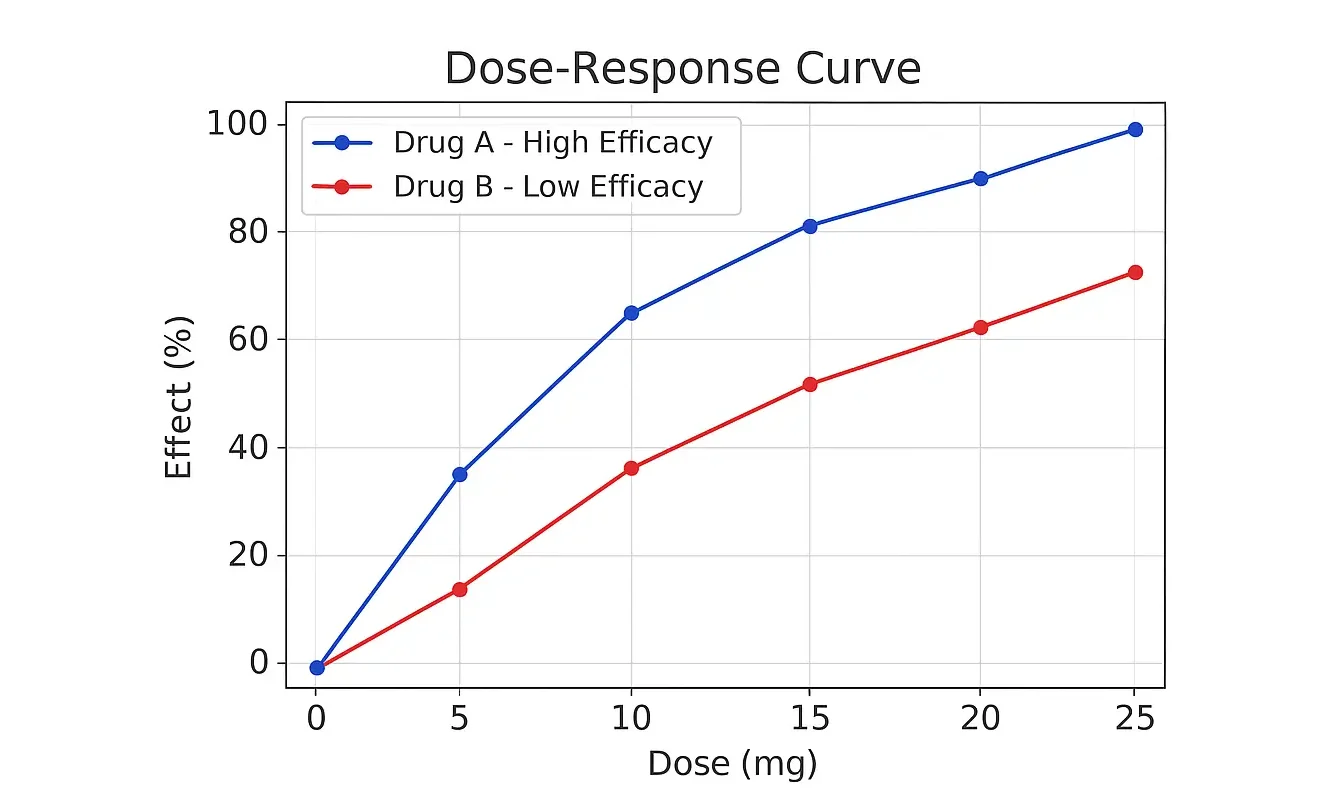
➔ Efficacy is about the maximum effect a drug can have, while potency is about how much drug you need to achieve a certain effect.
➢ Drug A: High efficacy (can reduce pain by 90%) but low potency (needs 20 mg to reduce pain by 50%).
➢ Drug B: Low efficacy (can only reduce pain by 50%) but high potency (needs only 5 mg to reduce pain by 50%).
➔ Efficacy and Potency are Independent: A drug can be highly potent (needs a small dose to work) but has low efficacy (doesn’t work very well even at high doses), and vice versa.

How it is tested:
➔ A researcher is testing two new antihypertensive drugs, Drug X and Drug Y, in a lab. The dose-response curves show:
- Drug X: Requires a low dose to reduce blood pressure but has a limited maximum effect.
- Drug Y: Requires a higher dose to reduce blood pressure but achieves a greater maximum reduction.
What’s true about this?
Answer: Drug X is more potent but less efficacious than Drug Y.
| Inhaled Anesthetic | Lipid Solubility (Potency – MAC↓) | Blood Solubility (Induction/Recovery) |
|---|---|---|
| Halothane | High (Very potent) | High (Slow induction/recovery) |
| Isoflurane | Moderate-High (Potent) | Moderate (Medium induction) |
| Sevoflurane | Moderate (Less potent) | Low (Fast induction/recovery) |
| Desflurane | Low (Least potent) | Very Low (Very fast induction) |
| Nitrous Oxide | Very Low (Least potent) | Very Low (Fastest induction) |
📝 Trick to remember:
◆ “High Lipid = High Potency = Low MAC”
◆ “High Blood = Slow Induction”
How it is tested:
A 55-year-old man undergoes general anesthesia for an elective abdominal surgery. Shortly after induction with an inhaled anesthetic, he develops hypotension and bradycardia. His past medical history is significant for liver disease. The anesthesiologist notes that the patient’s recovery is prolonged compared to expected. Which inhaled anesthetic was most likely used?
Answer: Halothane is metabolized in the liver and can cause halothane-induced hepatotoxicity. Hypotension & bradycardia are classic halothane side effects
Clearance and Half-life
➢ Clearance determines the rate at which a drug is eliminated, it influences the drug’s half-life (t₁/₂).
t½ = (0.693 × Vd) / CL
Key takeaway:
✔ ↑ Vd → ↑ t₁/₂
✔ ↑ CL → ↓ t₁/₂
➔ Impaired liver or kidney function → ↓ clearance → ↑ half-life → prolonged drug exposure → increased toxicity risk.
How it is tested:
➔ A 65-year-old woman with liver cirrhosis is prescribed a medication that is extensively metabolized by the liver. She later presents with elevated drug levels and signs of toxicity. What pharmacokinetic change explains this?
Answer: Liver cirrhosis → ↓ hepatic metabolism→ ↑ half-life → drug accumulation → toxicity.
Loading and Maintenance dose
Loading dose = (Cp × Vd) / F
How it is tested:
➔ A 68-year-old obese patient with pneumonia requires immediate antibiotic therapy. How does an increased volume of distribution (Vd) affect the required loading dose?
Answer: An increased Vd means the drug distributes more widely in the body, so a higher loading dose is required to reach the desired plasma concentration.
Maintenance dose = (Cp × CL × τ) / F
➔ Higher clearance (CL) → Faster elimination → Higher maintenance dose needed
➔ Longer dosing interval (τ) → More drug required per dose
Key takeaway:
✔ Adjust the loading dose based on Vd and the maintenance dose based on CL to ensure proper drug levels and avoid toxicity.
Drug metabolism
| Metabolism Phase | Reactions | Mnemonic | Key Clinical Point |
|---|---|---|---|
| Phase I (CYP450-dependent) |
| “Old Rats Hide” | Geriatrics lose Phase I first → “Old People Fade First” |
| Phase II (Conjugation) |
| “Mother Goose Ate Soup” | Phase II mostly intact in elderly → “2nd Chance Metabolism” |
How it is tested:
➔ A 78-year-old man with generalized anxiety disorder is prescribed diazepam. A few days later, he developed excessive drowsiness and confusion. His liver function tests are normal. Metabolic reaction most likely to be affected? Answer: Oxidation
| CYP-450 Inhibitors (“SICKFACES.COM”) |
|---|
| Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole |
| CYP-450 Inducers (“CRAP GPS”) |
|---|
| Carbamazepine, Rifampin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, St. John’s Wort |
Slow and Fast Acetylators
Slow acetylators struggle with these drugs. “SHIP” .These drugs can cause drug-induced lupus and positive for anti-histone ab
- Sulfasalazine
- Hydralazine
- Isoniazid
- Procainamide
How it is tested:
➔ A 55-year-old man with hypertension and hyperlipidemia is started on hydralazine, hydrochlorothiazide, and atorvastatin. Genetic testing confirms he is a slow acetylator. Drugs likely to be affected?
Answer: Hydralazine
Key takeaway:
✔ Slow acetylators → More drug accumulation → Higher toxicity risk
✔ Fast acetylators → Rapid clearance → Lower drug efficacy
Drug effect modifications
| Effect | Definition | Example |
|---|---|---|
| Additive | Combined effect = Sum (2 + 2 = 4) | Aspirin + Acetaminophen |
| Permissive | One drug enables another’s effect | Cortisol & Catecholamines |
| Synergistic | Combined effect > Sum (2 + 2 > 4) | Clopidogrel + Aspirin |
| Potentiation | Inactive drug boosts another (2 + 0 > 2) | Carbidopa + Levodopa |
| Antagonistic | Combined effect < Sum (2 + 2 < 4) | Ethanol for Methanol |
| Tachyphylaxis | Rapid tolerance with repeated use | Nitrates |
How it is tested:
A 45-year-old factory worker develops recurrent headaches, dizziness, and palpitations after repeated exposure to workplace chemicals. His symptoms gradually diminish with continued exposure but reappear after a brief break. What phenomenon explains this?
Answer: Tachyphylaxis (Rapid tolerance)
Autonomic receptors
Parasympathetic:
➔ Long preganglionic → ACh (Nn) → Short postganglionic → ACh (M)
Targets: Smooth muscle, glands, cardiac muscle
Sympathetic:
➔ Short preganglionic → ACh (Nn) → Long postganglionic → (variable)
➢ Most organs: NE (α1, α2, β1)
➢ Renal vasculature: Dopamine (D1)
➢ Sweat glands (EXCEPTION): ACh (M)
➢ Adrenal medulla: ACh (Nn) → Epi, NE (into blood)
➢ Somatic: ACh (Nm) → Skeletal muscle (direct, no ganglion)
Key takeaway:
✔ Sweat glands = Sympathetic but use ACh instead of norepinephrine.
✔ Adrenal medulla = Direct preganglionic innervation
How it is tested:
➔ A 42-year-old man presents with heat intolerance and inability to sweat despite exposure to high temperatures. Neurotransmitter responsible for impaired sweating?
Answer: Acetylcholine
G-Protein Coupled Receptors (GPCRs)
1. Gq-Coupled Receptors (↑ IP3/DAG → ↑ Ca²⁺ → Smooth Muscle Contraction)
Mnemonic: “Cutie HAVe 1 M&M”
(Cutie = “Q” for Gq)
- H1 (Histamine 1)
- A1 (Alpha-1 Adrenergic)
- V1 (Vasopressin 1)
- M1 (Muscarinic 1)
- M3 (Muscarinic 3)
2. Gs-Coupled Receptors (↑ cAMP → Activate PKA → Smooth Muscle Relaxation)
📌 Mnemonic: “Gs = Big, Dumb, Happy Vibes” 😃💨
- B1 (Beta-1 Adrenergic)
- B2 (Beta-2 Adrenergic)
- D1 (Dopamine 1)
- H2 (Histamine 2)
- V2 (Vasopressin 2)
3. Gi-Coupled Receptors (↓ cAMP → Inhibits PKA → Inhibitory Effects)
📌 Mnemonic: “Gi = MAD 2” 😡
- M2 (Muscarinic 2)
- A2 (Alpha-2 Adrenergic)
- D2 (Dopamine 2)
Adrenergic Receptors: Key Drug Targets
🟢= Agonist 🔴= Antagonist
➢ α₁: Vascular/visceral smooth muscle → Vasoconstriction.
Drugs: 🔴Tamsulosin (BPH), 🔴Prazosin (hypertension)
➢ α₂: Presynaptic → Dec sympathetic outflow
Drugs: 🟢Clonidine (HTN)
➢ β₁: Heart/kidney → ↑ HR
Drugs: 🔴Metoprolol (Rate control), 🟢Dobutamine (heart failure)
➢ β₂: Lungs → Bronchodilation
Drugs: 🟢Albuterol (asthma)
➢ β₃: Urinary bladder → Relaxes detrusor muscle
Drugs: 🟢Mirabegron (Urgency incontinence).
Key takeaway:
✔ Most Targeted: α₁, β₁, β₂.
✔ Blockers relax muscles (α₁), slows heart (β₁).
✔ Activators boost heart (β₁), open airways (β₂).
How it is tested:
➢ A 70-year-old man with BPH is prescribed tamsulosin to improve urinary flow. Which receptor does this drug target? And side effects?
Answer: Blocks α₁–adrenergic receptors, it can cause orthostatic hypotension.
➢ A 45-year-old asthmatic presents with acute wheezing and receives a nebulized medication for rapid relief. Which receptor does this drug target, and what is the effect?
Answer: Stimulates β₂-adrenergic receptors → bronchodilation.
Cholinergic Receptors: Key Drug Targets
➢ M₁: CNS, enteric nerves → Cognition, gut motility.
Drugs: 🟢Donepezil, Rivastigmine (Alzheimer’s)
➢ M₂: Heart → ↓ HR,
Drugs: 🔴Atropine (bradycardia)
➢ M₃: Glands, smooth muscle → Secretion, gut/ detrusor contraction, bronchoconstriction, miosis.
Drugs: 🟢Bethanechol (post-op urinary retention)
🔴Ipratropium (COPD, asthma)
🔴Oxybutynin (overactive bladder)
Key takeaway:
✔ Most Targeted: M₃ (glands, smooth muscle).
✔ for secretions (pilocarpine), cognition (donepezil), or bladder emptying (bethanechol).
✔ Block for overactive bladder (oxybutynin) or bronchospasm (ipratropium).
How it is tested:
➢ A 65-year-old woman undergoes a hysterectomy under general anesthesia. Post-operatively, she complains of an inability to urinate and has a palpable, distended bladder. DOC for this condition?
Answer: Bethanechol (M₃ agonist) → activates detrusor muscle → promotes bladder emptying.
➢ A 35-year-old farmer is brought to the emergency department with excessive salivation, lacrimation, and severe bradycardia (HR 38 bpm). He was exposed to organophosphate. Drug of choice?
Answer: Atropine (M antagonist) → blocks muscarinic receptors → increases heart rate.
Dopamine Receptors: Key Drug Targets
➢ D₁: Renal vasculature, CNS (striatum) → Vasodilation, activates direct pathway.
Drugs: 🟢Fenoldopam (hypertensive crisis), 🟢Levodopa – dopamine precursor (Parkinson’s).
➢ D₂: CNS (striatum, pituitary), presynaptic terminals → Modulates transmitter release, inhibits indirect pathway.
Drugs: 🔴Metoclopramide (antiemetic), 🔴Haloperidol (antipsychotic),
🟢Bromocriptine (hyperprolactinemia).
Key takeaway:
✔ Most Targeted: D₂ (CNS modulation).
✔ Stimulate for Parkinson’s (levodopa).
✔ Block for psychosis (haloperidol) or nausea (metoclopramide).
How it is tested:
➔ A 28-year-old man is brought to the emergency department with hallucinations, delusions, and aggressive behavior. He is given haloperidol. How does this drug work?
Answer: Haloperidol = D₂ blocker → treats acute psychosis by modulating dopamine signaling.
➔ A 45-year-old woman undergoing chemotherapy for breast cancer develops severe nausea and vomiting. She is prescribed domperidone to manage her symptoms. MAO of this drug?
Answer: Domperidone = D₂ blocker → treats chemotherapy-induced nausea/vomiting by inhibiting dopamine signaling in the CTZ.
Histamine Receptors: Key Drug Targets
➢ H₁: Nasal/bronchial mucosa → Mucus production, bronchoconstriction, pruritus.
Drugs: 🔴Diphenhydramine, 🔴Loratadine (allergies, itching).
➢ H₂: Stomach → Gastric acid secretion.
Drugs: 🔴Ranitidine, 🔴Famotidine (GERD, peptic ulcers).
Vasopressin Receptors: Key Drug Targets
➢ V₁: Vascular smooth muscle → Vasoconstriction.
Drugs: 🟢Vasopressin (enuresis).
➢ V₂: Kidney collecting tubules → Water reabsorption (via aquaporin-2).
Drugs: 🟢Desmopressin (DI), 🔴Tolvaptan (SIADH).
Key takeaway:
✔ H₁ blockers: Treat allergies, and itching.
✔ H₂ blockers: Reduce gastric acid.
✔ V₁ agonists: Vasoconstriction in shock.
✔ V₂ agonists/antagonists: Manage water balance (DI/SIADH).
How it is tested:
➔ A 25-year-old woman presents with sneezing, runny nose, and itchy eyes during the pollen season. She is treated with loratadine, and how does it work?
Answer: H₁ receptor antagonist → blocks histamine effects → reduces allergic symptoms (itching, mucus production).
➔ A 50-year-old man with a history of head trauma presents with excessive urination and thirst. Lab tests show dilute urine and hypernatremia. Which drug should be given, and how does it work?
Answer: Desmopressin (V₂ agonist) → increases water reabsorption in kidneys → treats central diabetes insipidus.
Drugs Acting in Autonomic Pathways
➔ Cholinergic Pathway
| Drug | Mechanism | Use |
|---|---|---|
| Botulinum Toxin | Blocks Acetylcholine (ACh) release – prevents muscle contraction. | Muscle spasm, cosmetic. |
| AChE Inhibitors (Neostigmine, Donepezil) | Inhibits Acetylcholine (ACh) breakdown – Inc ACh at synapse. | Myasthenia gravis, Alzheimer’s. |
➔ Noradrenergic Pathway
| Drug | Mechanism | Use |
|---|---|---|
| Amphetamines | Reverse NET (Norepinephrine Transporter) – Inc Norepinephrine (NE) release. | ADHD, narcolepsy. |
| NET Inhibitors (Cocaine, TCAs) | Block NE reuptake – Inc NE in synapse. | Cocaine (abuse), TCAs (depression). |
| a2 Agonists (Clonidine) | Stimulate presynaptic a2 receptors – Dec NE release. | Hypertension, ADHD, PTSD |
Organophosphate Poisoning
- Cause: Inhibition of acetylcholinesterase → Excess acetylcholine → Cholinergic toxicity
- Clinical Features (DUMBBELLS): Diarrhea, Urination, Miosis, Bradycardia, Bronchospasm, Bronchorrhea, Emesis, Lacrimation, Lethargy, Salivation, Sweating
- Treatment:
1. Atropine (muscarinic antagonist)
2. Pralidoxime (2-PAM) (reactivates acetylcholinesterase)
How it is tested:
→ A 10-year-old boy is brought to the clinic by his parents due to difficulty focusing in school, hyperactivity, and impulsivity. He was diagnosed with ADHD and started on methylphenidate. How does it work?
Answer: It Reverses NET → increases norepinephrine (NE) and dopamine release in the brain → improves focus and reduces hyperactivity.
→ A 45-year-old woman with a history of depression is taking a monoamine oxidase inhibitor (MAOI). She presents to the emergency department with severe hypertension, headache, and sweating after using cocaine at a party. Why did her symptoms worsen significantly compared to someone not on an MAOI?
Answer: Cocaine blocks NET/DAT → ↑ NE/DA in the synapse.
MAOIs inhibit MAO → prevent the breakdown of NE/DA → massive accumulation of NE/DA → severe hypertensive crisis (due to excessive vasoconstriction).
Physiologic effects and uses of sympathomimetics
- Norepinephrine (α > β): Causes vasoconstriction (α1) → ↑ Mean arterial pressure
(MAP), ↑ systolic & diastolic BP → reflex bradycardia. DOC for Cardiogenic shock and septic shock. - Epinephrine (α ≈ β): Balances vasoconstriction (α1) and vasodilation (β2) → ↑ HR (β1), moderate BP increase. DOC for Anaphylaxis
- Isoproterenol (β1 = β2, no α effects)
How it is tested:
→ A 72-year-old man with a history of coronary artery disease and decompensated heart failure is admitted to the ICU. His BP is 75/40 mmHg and his HR is 110 bpm, has cold and clammy extremities. Vasopressor of choice?
Answer: Norepinephrine (α1 > β1) is first-line for cardiogenic shock
Beers Criteria
➔ Reduces inappropriate prescribing in the elderly.
Common drugs to avoid in elderly due to increased risk of adverse events.
1) α-blockers: ↑ hypotension.
2) Anticholinergics, antidepressants, antihistamines, opioids: ↑ delirium, sedation, falls, constipation, urinary retention.
3) Benzodiazepines: ↑ delirium, sedation, falls.
4) NSAIDs: ↑ GI bleeding (worse with anticoagulants).
5) PPIs: ↑ C. difficile infection risk.
Age-Related Pharmacokinetic Shifts
A. Metabolism: ↓ hepatic mass, blood flow, drug metabolism. Phase I (e.g., diazepam) declines first; Phase II (e.g., lorazepam) is safer.
B. Absorption: ↑ gastric pH, ↓ gastric emptying; affected by drug/food interactions.
C. Distribution: ↑ body fat (↑ Vd lipophilic, e.g., propofol), ↓ albumin (↓ acidic drug binding), ↓ total body water (↓ Vd hydrophilic, e.g., digoxin).
D. Elimination: ↓ GFR, tubular secretion; ↓ doses for renally excreted drugs in elderly.
How it is tested:
→ A 78-year-old man with heart failure on digoxin presents with nausea, fatigue, confusion, and visual halos. His renal function is mildly impaired (eGFR 50 mL/min), but his serum digoxin level is significantly elevated. He has low total body water due to aging-related physiological changes. Cause of toxicity?
Answer: Low volume of distribution (Vd) in the elderly
If you need guidance on your target USMLE scores or have questions on improving your chances of matching into your dream specialty, feel free to schedule a call here or text us on WhatsApp here for personalized support.
🍀 We wish you best of luck on your exam!


2026 Residency Personal Statement The Ultimate Guide to Crafting Your Residency Personal Statement

Best Resources for USMLE STEP 1! How to Study and Prepare for STEP1?

ERAS Application 2026 Guide! How to Fill Out ERAS Residency Application?
About TheMatchGuy
RECOMMENDED ARTICLES

Best Electives in the US

Best Resources for USMLE STEP 1! How to Study and Prepare for STEP1?

United States Clinical Experience for IMGs